What Is the Optimal Timing of Transplantation of Neural Stem Cells in Spinal Cord Injury? A Systematic Review and Network Meta-Analysis Based on Animal Studies
- PMID: 35371014
- PMCID: PMC8965614
- DOI: 10.3389/fimmu.2022.855309
What Is the Optimal Timing of Transplantation of Neural Stem Cells in Spinal Cord Injury? A Systematic Review and Network Meta-Analysis Based on Animal Studies
Abstract
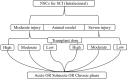
Objective: The optimal transplantation timing of neural stem cells in spinal cord injury is fully explored in animal studies to reduce the risk of transformation to clinical practice and to provide valuable reference for future animal studies and clinical research.
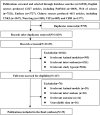
Method: Seven electronic databases, namely, PubMed, Web of Science, Embase, Wanfang, Chinese Scientific Journal Database (CSJD-VIP), China Biomedical Literature Database (CBM), and China National Knowledge Infrastructure (CNKI), were searched. The studies were retrieved from inception to November 2021. Two researchers independently screened the literature, extracted data, and evaluated the methodological quality based on the inclusion criteria.
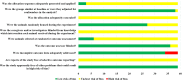
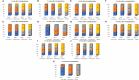
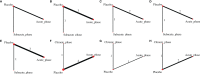
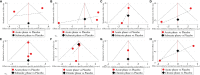
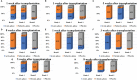
Results and discussion: Thirty-nine studies were incorporated into the final analyses. Based on the subgroup of animal models and transplantation dose, the results of network meta-analysis showed that the effect of transplantation in the subacute phase might be the best. However, the results of traditional meta-analysis were inconsistent. In the moderate-dose group of moderate spinal cord injury model and the low-dose group of severe spinal cord injury model, transplantation in the subacute phase did not significantly improve motor function. Given the lack of evidence for direct comparison between different transplantation phases, the indirectness of our network meta-analysis, and the low quality of evidence in current animal studies, our confidence in recommending cell transplantation in the subacute phase is limited. In the future, more high-quality, direct comparative studies are needed to explore this issue in depth.
Keywords: animal study; network meta-analysis; neural stem cells; spinal cord injury; systematic review; transplantation timing.
Copyright © 2022 Shang, Li, Chen, Wang, Wang, Zhang, Wang and Wanyan.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures



References
-
- GBD 2016 Traumatic Brain Injury and Spinal Cord Injury Collaborators . Global, Regional, and National Burden of Traumatic Brain Injury and Spinal Cord Injury, 1990-2016: A Systematic Analysis for the Global Burden of Disease Study 2016. Lancet Neurol (2019) 18:56–87. doi: 10.1016/S1474-4422(18)30415-0 - DOI - PMC - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

