Intermediate Cells of Dual Embryonic Origin Follow a Basal to Apical Gradient of Ingression Into the Lateral Wall of the Cochlea
- PMID: 35372344
- PMCID: PMC8964366
- DOI: 10.3389/fcell.2022.867153
Intermediate Cells of Dual Embryonic Origin Follow a Basal to Apical Gradient of Ingression Into the Lateral Wall of the Cochlea
Abstract
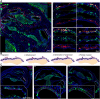
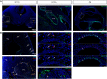
Intermediate cells of the stria vascularis are neural crest derived melanocytes. They are essential for the establishment of the endocochlear potential in the inner ear, which allows mechanosensory hair cells to transduce sound into nerve impulses. Despite their importance for normal hearing, how these cells develop and migrate to their position in the lateral wall of the cochlea has not been studied. We find that as early as E10.5 some Schwann cell precursors in the VIIIth ganglion begin to express melanocyte specific markers while neural crest derived melanoblasts migrate into the otic vesicle. Intermediate cells of both melanoblast and Schwann cell precursor origin ingress into the lateral wall of the cochlea starting at around E15.5 following a basal to apical gradient during embryonic development, and continue to proliferate postnatally.
Keywords: cochlea; inner ear; inner ear development; intermediate cells; melanoblast; neural crest; schwann cell precursor; stria vascularis.
Copyright © 2022 Renauld, Khan and Basch.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures



References
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

