Shuttle Transfer of mRNA Transcripts via Extracellular Vesicles From Male Reproductive Tract Cells to the Cumulus-Oocyte Complex in Rabbits (Oryctolagus cuniculus)
- PMID: 35372562
- PMCID: PMC8968341
- DOI: 10.3389/fvets.2022.816080
Shuttle Transfer of mRNA Transcripts via Extracellular Vesicles From Male Reproductive Tract Cells to the Cumulus-Oocyte Complex in Rabbits (Oryctolagus cuniculus)
Abstract
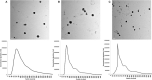
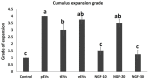
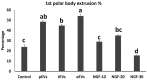
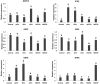
Semen is known to contain an ovulation-inducing factor (identified as a nerve growth factor, NGF) that shows a significant increase in ovulation after semen deposition in induced ovulatory species. However, the interplay between the male reproductive tract cells and oocyte maturation through messenger RNA (mRNA) cargo is yet to be investigated. Extracellular vesicles (EVs) from the primary culture of rabbit prostate (pEVs), epididymis (eEVs), and testis (tEVs) were isolated to examine their contents for several mRNA transcripts through relative quantitative PCR (RT-qPCR). The expressions of NGF, neurotrophin (NTF3), vascular endothelial growth factor A (VEGFA), A disintegrin and metalloprotease 17 (ADAM17), midkine (MDK), kisspeptin (KISS1), and gonadotrophin-releasing hormone (GNRH1) were examined in isolated EVs. EVs were characterized through transmission electron microscopy. EV uptake by cumulus cell culture was confirmed through microscopic detection of PKH26-stained EVs. Furthermore, the effects of pEVs, eEVs, and tEVs were compared with NGF (10, 20, and 30 ng/ml) supplementation on oocyte in vitro maturation (IVM) and transcript expression. KISS1, NTF3, MDK, ADAM17, GAPDH, and ACTB were detected in all EV types. GNRH1 was detected in tEVs. NGF was detected in pEVs, whereas VEGFA was detected in eEVs. pEVs, eEVs, and 20 ng/ml NGF showed the highest grade of cumulus expansion, followed by tEVs and 10 ng/ml NGF. Control groups and 30 ng/ml NGF showed the least grade of cumulus expansion. Similarly, first polar body (PB) extrusion was significantly increased in oocytes matured with eEVs, pEVs, tEVs, NGF20 (20 ng/ml NGF), NGF10 (10 ng/ml NGF), control, and NGF30 (30 ng/ml NGF). Additionally, the expression of NGFR showed a 1.5-fold increase in cumulus cells supplemented with eEVs compared with the control group, while the expression of PTGS2 (COX2) and NTRK showed 3-fold and 5-fold increase in NGF20-supplemented cumulus-oocyte complexes (COCs), respectively. Oocyte PMP15 expression showed a 1.8-fold increase in IVM medium supplemented with eEVs. Additionally, oocyte NGFR and NTRK expressions were drastically increased in IVM medium supplemented with pEVS (3.2- and 1.6-fold, respectively) and tEVs (4- and 1.7-fold, respectively). This is the first report to examine the presence of mRNA cargo in the EVs of male rabbit reproductive tract cells that provides a model for the stimulation of female rabbits after semen deposition.
Keywords: epididymis; extracellular vesicles; oocyte; prostate; rabbit; testis.
Copyright © 2022 Abumaghaid, Abdelazim, Belali, Alhujaily and Saadeldin.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures




References
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

