Potential Role of the Host-Derived Cell-Wall Binding Domain of Endolysin CD16/50L as a Molecular Anchor in Preservation of Uninfected Clostridioides difficile for New Rounds of Phage Infection
- PMID: 35377223
- PMCID: PMC9045149
- DOI: 10.1128/spectrum.02361-21
Potential Role of the Host-Derived Cell-Wall Binding Domain of Endolysin CD16/50L as a Molecular Anchor in Preservation of Uninfected Clostridioides difficile for New Rounds of Phage Infection
Abstract
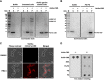
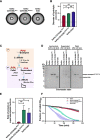
Endolysin is a phage-encoded cell-wall hydrolase which degrades the peptidoglycan layer of the bacterial cell wall. The enzyme is often expressed at the late stage of the phage lytic cycle and is required for progeny escape. Endolysins of bacteriophage that infect Gram-positive bacteria often comprises two domains: a peptidoglycan hydrolase and a cell-wall binding domain (CBD). Although the catalytic domain of endolysin is relatively well-studied, the precise role of CBD is ambiguous and remains controversial. Here, we focus on the function of endolysin CBD from a recently isolated Clostridioides difficile phage. We found that the CBD is not required for lytic activity, which is strongly prevented by the surface layer of C. difficile. Intriguingly, hidden Markov model analysis suggested that the endolysin CBD is likely derived from the CWB2 motif of C. difficile cell-wall proteins but possesses a higher binding affinity to bacterial cell-wall polysaccharides. Moreover, the CBD forms a homodimer, formation of which is necessary for interaction with the surface saccharides. Importantly, endolysin diffusion and sequential cytolytic assays showed that CBD of endolysin is required for the enzyme to be anchored to post-lytic cell-wall remnants, suggesting its physiological roles in limiting diffusion of the enzyme, preserving neighboring host cells, and thereby enabling the phage progeny to initiate new rounds of infection. Taken together, this study provides an insight into regulation of endolysin through CBD and can potentially be applied for endolysin treatment against C. difficile infection. IMPORTANCE Endolysin is a peptidoglycan hydrolase encoded in a phage genome. The enzyme is attractive due to its potential use as antibacterial treatment. To utilize endolysin for the therapeutic propose, understanding of the fundamental role of endolysin becomes important. Here, we investigate the function of cell-wall binding domain (CBD) of an endolysin from a C. difficile phage. The domain is homologous to a cell-wall associating module of bacterial cell-wall proteins, likely acquired during phage-host coevolution. The interaction of CBD to bacterial cell walls reduces enzyme diffusion and thereby limits cell lysis of the neighboring bacteria. Our findings indicate that the endolysin is trapped to the cell-wall residuals through CBD and might serve as an advantage for phage replication. Thus, employing a CBD-less endolysin might be a feasible strategy for using endolysin for the treatment of C. difficile infection.
Keywords: Clostridioides difficile; bacteria-phage coevolution; cell-wall binding domain; endolysin; peptidoglycan; surface layer.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



Similar articles
-
Molecular basis for recognition of the Group A Carbohydrate backbone by the PlyC streptococcal bacteriophage endolysin.Biochem J. 2021 Jun 25;478(12):2385-2397. doi: 10.1042/BCJ20210158. Biochem J. 2021. PMID: 34096588 Free PMC article.
-
Characterization of the host specificity of the SH3 cell wall binding domain of the staphylococcal phage 88 endolysin.Arch Microbiol. 2025 Jan 29;207(2):47. doi: 10.1007/s00203-025-04242-1. Arch Microbiol. 2025. PMID: 39878790
-
The multidomain architecture of a bacteriophage endolysin enables intramolecular synergism and regulation of bacterial lysis.J Biol Chem. 2021 Jan-Jun;296:100639. doi: 10.1016/j.jbc.2021.100639. Epub 2021 Apr 8. J Biol Chem. 2021. PMID: 33838182 Free PMC article.
-
Bacteriophage endolysins as a potential weapon to combat Clostridioides difficile infection.Gut Microbes. 2020 Nov 9;12(1):1813533. doi: 10.1080/19490976.2020.1813533. Gut Microbes. 2020. PMID: 32985336 Free PMC article. Review.
-
Bacteriophage and peptidoglycan degrading enzymes with antimicrobial applications.Recent Pat Biotechnol. 2007;1(2):113-22. doi: 10.2174/187220807780809463. Recent Pat Biotechnol. 2007. PMID: 19075835 Review.
Cited by
-
Emerging applications of phage therapy and fecal virome transplantation for treatment of Clostridioides difficile infection: challenges and perspectives.Gut Pathog. 2023 May 9;15(1):21. doi: 10.1186/s13099-023-00550-3. Gut Pathog. 2023. PMID: 37161478 Free PMC article. Review.
-
Phage-derived proteins: Advancing food safety through biocontrol and detection of foodborne pathogens.Compr Rev Food Sci Food Saf. 2025 Mar;24(2):e70124. doi: 10.1111/1541-4337.70124. Compr Rev Food Sci Food Saf. 2025. PMID: 39898971 Free PMC article. Review.
-
Characterization of Lactobacilli Phage Endolysins and Their Functional Domains-Potential Live Biotherapeutic Testing Reagents.Viruses. 2023 Sep 23;15(10):1986. doi: 10.3390/v15101986. Viruses. 2023. PMID: 37896764 Free PMC article.
-
New treatment approaches for Clostridioides difficile infections: alternatives to antibiotics and fecal microbiota transplantation.Gut Microbes. 2024 Jan-Dec;16(1):2337312. doi: 10.1080/19490976.2024.2337312. Epub 2024 Apr 9. Gut Microbes. 2024. PMID: 38591915 Free PMC article. Review.
-
The long and sinuous road to phage-based therapy of Clostridioides difficile infections.Front Med (Lausanne). 2023 Aug 23;10:1259427. doi: 10.3389/fmed.2023.1259427. eCollection 2023. Front Med (Lausanne). 2023. PMID: 37680620 Free PMC article. Review.
References
-
- Centers for Disease Control and Prevention. 2019. Antibiotic resistance threats in the United States, 2019. Centers for Disease Control and Prevention.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

