Monoclonal Antibodies in the Treatment of Relapsing Multiple Sclerosis: an Overview with Emphasis on Pregnancy, Vaccination, and Risk Management
- PMID: 35378683
- PMCID: PMC8978776
- DOI: 10.1007/s13311-022-01224-9
Monoclonal Antibodies in the Treatment of Relapsing Multiple Sclerosis: an Overview with Emphasis on Pregnancy, Vaccination, and Risk Management
Abstract
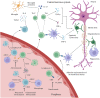
Monoclonal antibodies have become a mainstay in the treatment of patients with relapsing multiple sclerosis (RMS) and provide some benefit to patients with primary progressive MS. They are highly precise by specifically targeting molecules displayed on cells involved in distinct immune mechanisms of MS pathophysiology. They not only differ in the target antigen they recognize but also by the mode of action that generates their therapeutic effect. Natalizumab, an [Formula: see text]4[Formula: see text]1 integrin antagonist, works via binding to cell surface receptors, blocking the interaction with their ligands and, in that way, preventing the migration of leukocytes across the blood-brain barrier. On the other hand, the anti-CD52 monoclonal antibody alemtuzumab and the anti-CD20 monoclonal antibodies rituximab, ocrelizumab, ofatumumab, and ublituximab work via eliminating selected pathogenic cell populations. However, potential adverse effects may be serious and can necessitate treatment discontinuation. Most importantly, those are the risk for (opportunistic) infections, but also secondary autoimmune diseases or malignancies. Monoclonal antibodies also carry the risk of infusion/injection-related reactions, primarily in early phases of treatment. By careful patient selection and monitoring during therapy, the occurrence of these potentially serious adverse effects can be minimized. Monoclonal antibodies are characterized by a relatively long pharmacologic half-life and pharmacodynamic effects, which provides advantages such as permitting infrequent dosing, but also creates disadvantages regarding vaccination and family planning. This review presents an overview of currently available monoclonal antibodies for the treatment of RMS, including their mechanism of action, efficacy and safety profile. Furthermore, we provide practical recommendations for risk management, vaccination, and family planning.
Keywords: Alemtuzumab; Disease-modifying therapy; Monoclonal antibodies; Multiple sclerosis; Natalizumab; Ocrelizumab; Ofatumumab; Rituximab; Ublituximab.
© 2022. The Author(s).
Conflict of interest statement
Nik Krajnc has participated in meetings sponsored by, received speaker honoraria or travel funding from Roche, Novartis, and Merck and holds a grant for a Multiple Sclerosis Clinical Training Fellowship Programme from the European Committee for Treatment and Research in Multiple Sclerosis (ECTRIMS). Gabriel Bsteh has participated in meetings sponsored by, received speaker honoraria or travel funding from Biogen, Celgene, Lilly, Merck, Novartis, Sanofi-Genzyme, and Teva and received honoraria for consulting Biogen, Celgene, Novartis, Sanofi-Genzyme, Roche, and Teva. Thomas Berger has participated in meetings sponsored by and received honoraria (lectures, advisory boards, consultations) from pharmaceutical companies marketing treatments for MS: Allergan, Bayer, Biogen, Bionorica, Celgene, MedDay, Merck, Novartis, Octapharma, Roche, Sanofi-Genzyme, Teva. His institution has received financial support in the past 12 months by unrestricted research grants (Biogen, Bayer, Merck, Novartis, Sanofi Aventis, Teva and for participation in clinical trials in multiple sclerosis sponsored by Alexion, Bayer, Biogen, Merck, Novartis, Octapharma, Roche, Sanofi-Genzyme, Teva. Jan Mares declares no conflict of interest relevant to this manuscript. Hans-Peter Hartung has participated in meetings sponsored by and received honoraria (lectures, advisory boards, consultations) from pharmaceutical companies marketing treatments for MS: Alexion, Bayer, Biogen, Bristol Myers Squibb, GeNeuro, Horizon Therapeutics, Merck, Novartis, Octapharma, Roche, TG Therapeutics, UCB.
Figures
References
-
- Smith SL. Ten years of Orthoclone OKT3 (muromonab-CD3): a review. J Transpl Coord. 1996;6(3):109–19; quiz 120–1. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

