Inhibition of CXXC5 function reverses obesity-related metabolic diseases
- PMID: 35384342
- PMCID: PMC8982507
- DOI: 10.1002/ctm2.742
Inhibition of CXXC5 function reverses obesity-related metabolic diseases
Abstract
Background: Metabolic diseases, including type 2 diabetes, have long been considered incurable, chronic conditions resulting from a variety of pathological conditions in obese patients. Growing evidence suggests the Wnt/β-catenin pathway is a major pathway in adipose tissue remodelling, pancreatic β-cell regeneration and energy expenditure through regulation of key metabolic target genes in various tissues. CXXC5-type zinc finger protein 5 (CXXC5) is identified negative feedback regulator of the Wnt/β-catenin pathway that functions via Dishevelled (Dvl) binding.
Methods: Expression level of CXXC5 was characterised in clinical samples and diabetes-induced mice model. Diabetes-induced mice model was established by using high-fat diet (HFD). HFD-fed mice treated with KY19334, a small molecule inhibiting CXXC5-Dvl protein-protein interaction (PPI), was used to assess the role of CXXC5 in metabolic diseases.
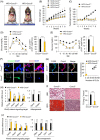
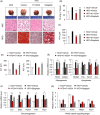
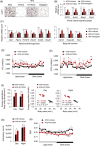
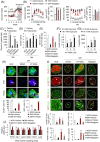
Results: Here, we show that CXXC5 is overexpressed with suppression of Wnt/β-catenin signalling in visceral adipose tissues of patients with obesity-related diabetes. Meanwhile, Cxxc5-/- mice fed an HFD exhibited resistance to metabolic dysregulation. KY19334 restores the lowered Wnt/β-catenin signalling and reverses metabolic abnormalities as observed in HFD-fed Cxxc5-/- mice. Administration of KY19334 on HFD-fed mice had a long-lasting glucose-controlling effect through remodelling of adipocytes and regeneration of pancreatic β-cells.
Conclusion: Overall, the inhibition of CXXC5 function by small molecule-mediated interference of Dvl binding is a potential therapeutic strategy for the treatment of obesity-related diabetes.
Keywords: CXXC5; Wnt/β-catenin pathway; adipose tissue remodelling; metabolic diseases; pancreatic β-cell regeneration.
© 2022 The Authors. Clinical and Translational Medicine published by John Wiley & Sons Australia, Ltd on behalf of Shanghai Institute of Clinical Bioinformatics.
Conflict of interest statement
The authors declare no competing interests exist.
Figures


References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
