Molecular and biological characterization of an Asian-American isolate of Chikungunya virus
- PMID: 35385544
- PMCID: PMC8985947
- DOI: 10.1371/journal.pone.0266450
Molecular and biological characterization of an Asian-American isolate of Chikungunya virus
Abstract
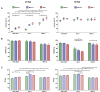
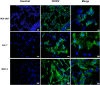
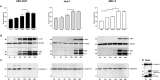
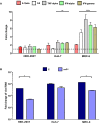
Chikungunya virus is an arthropod-transmitted virus that causes chikungunya fever, a disease characterized by severe muscle and joint pain. In 2013, the virus was introduced to the Americas and caused approximately 2.7 million cases of infection during the subsequent two years. The lack of knowledge regarding the biological behavior of the viral strains circulating during the outbreak motivated the characterization of an isolate from the Colombian outbreak, starting from analysis of the complete genome to the biological behavior in vitro. The full genome was retrieved using next-generation sequencing. The infective and replicative capacities were evaluated in HEK293T, Huh-7, and MRC-5 cell lines. The infection rates were determined by flow cytometry, and the cytopathic effect was assessed by a resazurin fluorescent metabolic assay. The viral yield was quantified using the virus plaque formation assay, while the viral proteins and genomic RNA kinetics were subsequently evaluated by western-blot and RT-qPCR. The COL7624 isolate clustered with other American and Caribbean sequences in the Asian American lineage. The T669A substitution in E2 protein distinguished it from other Colombian sequences reported in 2014. After 48 h post infection (hpi), the three cell lines analyzed reached infection percentages exceeding 65%, generating a high load of infectious viral progeny. The infection kinetics indicated that the replication peak of this CHIKV isolate is around 24 hpi, although gRNA is detectable in the culture supernatant from 4 hpi onwards. The infection caused the overexpression of interferon and pro-inflammatory cytokines, such as IL-1β, TNF-α, and IL-8. The COL7624 CHIKV isolate exhibited a high infective and replicative capacity as well as activation of cellular immune responses, similar to isolates belonging to the other genotypes.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures

References
-
- Economopoulou A, Dominguez M, Helynck B, Sissoko D, Wichmann O, Quenel P, et al.. Atypical Chikungunya virus infections: clinical manifestations, mortality and risk factors for severe disease during the 2005–2006 outbreak on Réunion. Epidemiol Infect. 2009;137: 534–541. doi: 10.1017/S0950268808001167 - DOI - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

