Biological Characteristics and Clinical Significance of Soluble PD-1/PD-L1 and Exosomal PD-L1 in Cancer
- PMID: 35386715
- PMCID: PMC8977417
- DOI: 10.3389/fimmu.2022.827921
Biological Characteristics and Clinical Significance of Soluble PD-1/PD-L1 and Exosomal PD-L1 in Cancer
Abstract
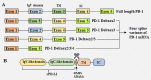
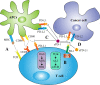
The immune checkpoint pathway consisting of the cell membrane-bound molecule programmed death protein 1 (PD-1) and its ligand PD-L1 has been found to mediate negative regulatory signals that effectively inhibit T-cell proliferation and function and impair antitumor immune responses. Considerable evidence suggests that the PD-1/PD-L1 pathway is responsible for tumor immune tolerance and immune escape. Blockage of this pathway has been found to reverse T lymphocyte depletion and restore antitumor immunity. Antagonists targeting this pathway have shown significant clinical activity in specific cancer types. Although originally identified as membrane-type molecules, several other forms of PD-1/PD-L1 have been detected in the blood of cancer patients, including soluble PD-1/PD-L1 (sPD-1/sPD-L1) and exosomal PD-L1 (exoPD-L1), increasing the composition and functional complications of the PD-1/PD-L1 signaling pathway. For example, sPD-1 has been shown to block the PD-1/PD-L immunosuppressive pathway by binding to PD-L1 and PD-L2, whereas the role of sPD-L1 and its mechanism of action in cancer remain unclear. In addition, many studies have investigated the roles of exoPD-L1 in immunosuppression, as a biomarker for tumor progression and as a predictive biomarker for response to immunotherapy. This review describes the molecular mechanisms underlying the generation of sPD-1/sPD-L1 and exoPD-L1, along with their biological activities and methods of detection. In addition, this review discusses the clinical importance of sPD-1/sPD-L1 and exoPD-L1 in cancer, including their predictive and prognostic roles and the effects of treatments that target these molecules.
Keywords: biological activity; cancer; efficacy prediction; exosomal PD-L1; immunotherapy; prognosis; soluble PD-1; soluble PD-L1.
Copyright © 2022 Niu, Liu, Yi, Jiao and Wu.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

