Antigen Uptake After Intradermal Microinjection Depends on Antigen Nature and Formulation, but Not on Injection Depth
- PMID: 35386985
- PMCID: PMC8974696
- DOI: 10.3389/falgy.2021.642788
Antigen Uptake After Intradermal Microinjection Depends on Antigen Nature and Formulation, but Not on Injection Depth
Abstract
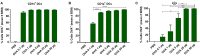
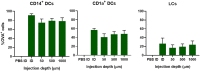
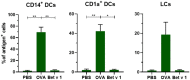
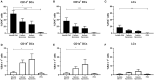
The skin is an attractive alternative administration route for allergy vaccination, as the skin is rich in dendritic cells (DCs) and is easily accessible. In the skin multiple subsets of DCs with distinct roles reside at different depths. In this study antigen (=allergen for allergy) formulations were injected in ex vivo human skin in a depth-controlled manner by using a hollow microneedle injection system. Biopsies were harvested at the injection site, which were then cultured for 72 h. Subsequently, the crawled-out cells were collected from the medium and analyzed with flow cytometry. Intradermal administration of ovalbumin (OVA, model antigen) solution at various depths in the skin did not affect the migration and maturation of DCs. OVA was taken up efficiently by the DCs, and this was not affected by the injection depth. In contrast, Bet v 1, the major allergen in birch pollen allergy, was barely taken up by dermal DCs (dDCs). Antigens were more efficiently taken up by CD14+ dDCs than CD1a+ dDCs, which in turn were more efficient at taken up antigen than Langerhans cells. Subsequently, both OVA and Bet v 1 were formulated in cationic and anionic liposomes, which altered antigen uptake drastically following intradermal microinjection. While OVA uptake was reduced by formulation in liposomes, Bet v 1 uptake in dDCs was increased by encapsulation in both cationic and anionic liposomes. This highlights the potential use of liposomes as adjuvant in intradermal allergy vaccine delivery. In conclusion, we observed that antigen uptake after intradermal injection was not affected by injection depth, but varied between different antigens and formulation.
Keywords: Bet v 1; allergen specific immunotherapy; antigen uptake; injection depth; intradermal injection; liposomes; ovalbumin.
Copyright © 2021 Leboux, Schipper, van Capel, Kong, van der Maaden, Kros, Jiskoot, de Jong and Bouwstra.
Conflict of interest statement
KvdM was employed by the company uPRAX. WJ is a scientific advisor at Coriolis Pharma, Martinsried, Germany. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
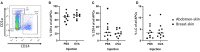


References
-
- Klimek L, Brehler R, Hamelmann E, Kopp M, Ring J, Treudler R, et al. Development of subcutaneous allergen immunotherapy (part 2): preventive aspects and innovations. Allergo J Int. (2019) 28:107–19. 10.1007/s40629-019-0097-z - DOI
-
- Bonnotte B, Gough M, Phan V, Ahmed A, Chong H, Martin F, et al. Intradermal injection, as opposed to subcutaneous injection, enhances immunogenicity and suppresses tumorigenicity of tumor cells. Cancer Res. (2003) 63:2145–9. - PubMed
LinkOut - more resources
Full Text Sources
Research Materials

