Iron Dyshomeostasis and Ferroptosis: A New Alzheimer's Disease Hypothesis?
- PMID: 35391749
- PMCID: PMC8981915
- DOI: 10.3389/fnagi.2022.830569
Iron Dyshomeostasis and Ferroptosis: A New Alzheimer's Disease Hypothesis?
Abstract
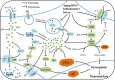
Iron plays a crucial role in many physiological processes of the human body, but iron is continuously deposited in the brain as we age. Early studies found iron overload is directly proportional to cognitive decline in Alzheimer's disease (AD). Amyloid precursor protein (APP) and tau protein, both of which are related to the AD pathogenesis, are associated with brain iron metabolism. A variety of iron metabolism-related proteins have been found to be abnormally expressed in the brains of AD patients and mouse models, resulting in iron deposition and promoting AD progression. Amyloid β (Aβ) and hyperphosphorylated tau, two pathological hallmarks of AD, can also promote iron deposition in the brain, forming a vicious cycle of AD development-iron deposition. Iron deposition and the subsequent ferroptosis has been found to be a potential mechanism underlying neuronal loss in many neurodegenerative diseases. Iron chelators, antioxidants and hepcidin were found useful for treating AD, which represents an important direction for AD treatment research and drug development in the future. The review explored the deep connection between iron dysregulation and AD pathogenesis, discussed the potential of new hypothesis related to iron dyshomeostasis and ferroptosis, and summarized the therapeutics capable of targeting iron, with the expectation to draw more attention of iron dysregulation and corresponding drug development.
Keywords: Alzheimer’s disease; FPN1; GPX4; ferroptosis; hepcidin; iron; iron chelator; lipid peroxidation.
Copyright © 2022 Wang, Wang, Shen, Li, Rausch and Huang.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
Similar articles
-
Revisiting the intersection of microglial activation and neuroinflammation in Alzheimer's disease from the perspective of ferroptosis.Chem Biol Interact. 2023 Apr 25;375:110387. doi: 10.1016/j.cbi.2023.110387. Epub 2023 Feb 8. Chem Biol Interact. 2023. PMID: 36758888 Review.
-
Iron dyshomeostasis and ferroptosis in Alzheimer's disease: Molecular mechanisms of cell death and novel therapeutic drugs and targets for AD.Front Pharmacol. 2022 Sep 16;13:983623. doi: 10.3389/fphar.2022.983623. eCollection 2022. Front Pharmacol. 2022. PMID: 36188557 Free PMC article. Review.
-
Iron dyshomeostasis, lipid peroxidation and perturbed expression of cystine/glutamate antiporter in Alzheimer's disease: Evidence of ferroptosis.Redox Biol. 2020 May;32:101494. doi: 10.1016/j.redox.2020.101494. Epub 2020 Mar 5. Redox Biol. 2020. PMID: 32199332 Free PMC article.
-
Iron and Targeted Iron Therapy in Alzheimer's Disease.Int J Mol Sci. 2023 Nov 15;24(22):16353. doi: 10.3390/ijms242216353. Int J Mol Sci. 2023. PMID: 38003544 Free PMC article. Review.
-
Ferroptosis in Alzheimer's Disease: The Regulatory Role of Glial Cells.J Integr Neurosci. 2025 Apr 24;24(4):25845. doi: 10.31083/JIN25845. J Integr Neurosci. 2025. PMID: 40302253 Review.
Cited by
-
Increased Expression of Transferrin Receptor 1 in the Brain Cortex of 5xFAD Mouse Model of Alzheimer's Disease Is Associated with Activation of HIF-1 Signaling Pathway.Mol Neurobiol. 2024 Sep;61(9):6383-6394. doi: 10.1007/s12035-024-03990-3. Epub 2024 Feb 1. Mol Neurobiol. 2024. PMID: 38296900 Free PMC article.
-
New Possibilities in the Therapeutic Approach to Alzheimer's Disease.Int J Mol Sci. 2022 Aug 10;23(16):8902. doi: 10.3390/ijms23168902. Int J Mol Sci. 2022. PMID: 36012193 Free PMC article. Review.
-
Alterations in zinc, copper, and iron levels in the retina and brain of Alzheimer's disease patients and the APP/PS1 mouse model.Metallomics. 2024 Dec 2;16(12):mfae053. doi: 10.1093/mtomcs/mfae053. Metallomics. 2024. PMID: 39520546 Free PMC article.
-
The role of ferroptosis in Alzheimer's disease: Mechanisms and therapeutic potential (Review).Mol Med Rep. 2025 Jul;32(1):192. doi: 10.3892/mmr.2025.13557. Epub 2025 May 9. Mol Med Rep. 2025. PMID: 40341407 Free PMC article. Review.
-
Phytochemicals as modulators of ferroptosis: a novel therapeutic avenue in cancer and neurodegeneration.Mol Biol Rep. 2025 Jun 25;52(1):636. doi: 10.1007/s11033-025-10752-4. Mol Biol Rep. 2025. PMID: 40560244 Review.
References
-
- Abdul Y., Li W., Ward R., Abdelsaid M., Hafez S., Dong G., et al. (2021). Deferoxamine Treatment Prevents Post-Stroke Vasoregression and Neurovascular Unit Remodeling Leading to Improved Functional Outcomes in Type 2 Male Diabetic Rats: Role of Endothelial Ferroptosis. Transl. Stroke Res. 12 615–630. 10.1007/s12975-020-00844-7 - DOI - PMC - PubMed
Publication types
LinkOut - more resources
Full Text Sources

