Long-term non-invasive drug treatments in adult zebrafish that lead to melanoma drug resistance
- PMID: 35394030
- PMCID: PMC9118090
- DOI: 10.1242/dmm.049401
Long-term non-invasive drug treatments in adult zebrafish that lead to melanoma drug resistance
Abstract
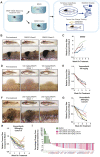
Zebrafish embryos are widely used for drug discovery, however, administering drugs to adult zebrafish is limited by current protocols that can cause stress. Here, we developed a drug formulation and administration method for adult zebrafish by producing food-based drug pellets that are consumed voluntarily. We applied this to zebrafish with BRAF-mutant melanoma, a model that has significantly advanced our understanding of melanoma progression, but not of drug resistance due to the limitations of current treatment methods. Zebrafish with melanomas responded to short-term, precise and daily dosing with drug pellets made with the BRAFV600E inhibitor, vemurafenib. On-target drug efficacy was determined by phospho-Erk staining. Continued drug treatment led to the emergence, for the first time in zebrafish, of acquired drug resistance and melanoma relapse, modelling the responses seen in melanoma patients. This method presents a controlled, non-invasive approach that permits long-term drug studies and can be widely applied to adult zebrafish models.
Keywords: Adult zebrafish; Drug delivery; Drug resistance; Long-term treatment; Melanoma.
© 2022. Published by The Company of Biologists Ltd.
Conflict of interest statement
Competing interests E.E.P. is the Editor-in-Chief at Disease Models & Mechanisms but was not included in any aspect of the editorial handling of this article or peer review process.
Figures



References
-
- Al-Olabi, L., Polubothu, S., Dowsett, K., Andrews, K. A., Stadnik, P., Joseph, A. P., Knox, R., Pittman, A., Clark, G., Baird, W.et al. (2018). Mosaic RAS/MAPK variants cause sporadic vascular malformations which respond to targeted therapy. J. Clin. Invest. 128, 5185. 10.1172/JCI124649 - DOI - PMC - PubMed
-
- Ceol, C. J., Houvras, Y., Jane-Valbuena, J., Bilodeau, S., Orlando, D. A., Battisti, V., Fritsch, L., Lin, W. M., Hollmann, T. J., Ferre, F.et al. (2011). The histone methyltransferase SETDB1 is recurrently amplified in melanoma and accelerates its onset. Nature 471, 513-517. 10.1038/nature09806 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

