The Potential of Current Noninvasive Wearable Technology for the Monitoring of Physiological Signals in the Management of Type 1 Diabetes: Literature Survey
- PMID: 35394448
- PMCID: PMC9034434
- DOI: 10.2196/28901
The Potential of Current Noninvasive Wearable Technology for the Monitoring of Physiological Signals in the Management of Type 1 Diabetes: Literature Survey
Abstract
Background: Monitoring glucose and other parameters in persons with type 1 diabetes (T1D) can enhance acute glycemic management and the diagnosis of long-term complications of the disease. For most persons living with T1D, the determination of insulin delivery is based on a single measured parameter-glucose. To date, wearable sensors exist that enable the seamless, noninvasive, and low-cost monitoring of multiple physiological parameters.
Objective: The objective of this literature survey is to explore whether some of the physiological parameters that can be monitored with noninvasive, wearable sensors may be used to enhance T1D management.
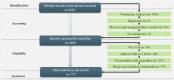
Methods: A list of physiological parameters, which can be monitored by using wearable sensors available in 2020, was compiled by a thorough review of the devices available in the market. A literature survey was performed using search terms related to T1D combined with the identified physiological parameters. The selected publications were restricted to human studies, which had at least their abstracts available. The PubMed and Scopus databases were interrogated. In total, 77 articles were retained and analyzed based on the following two axes: the reported relations between these parameters and T1D, which were found by comparing persons with T1D and healthy control participants, and the potential areas for T1D enhancement via the further analysis of the found relationships in studies working within T1D cohorts.
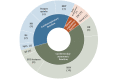
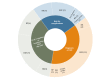
Results: On the basis of our search methodology, 626 articles were returned, and after applying our exclusion criteria, 77 (12.3%) articles were retained. Physiological parameters with potential for monitoring by using noninvasive wearable devices in persons with T1D included those related to cardiac autonomic function, cardiorespiratory control balance and fitness, sudomotor function, and skin temperature. Cardiac autonomic function measures, particularly the indices of heart rate and heart rate variability, have been shown to be valuable in diagnosing and monitoring cardiac autonomic neuropathy and, potentially, predicting and detecting hypoglycemia. All identified physiological parameters were shown to be associated with some aspects of diabetes complications, such as retinopathy, neuropathy, and nephropathy, as well as macrovascular disease, with capacity for early risk prediction. However, although they can be monitored by available wearable sensors, most studies have yet to adopt them, as opposed to using more conventional devices.
Conclusions: Wearable sensors have the potential to augment T1D sensing with additional, informative biomarkers, which can be monitored noninvasively, seamlessly, and continuously. However, significant challenges associated with measurement accuracy, removal of noise and motion artifacts, and smart decision-making exist. Consequently, research should focus on harvesting the information hidden in the complex data generated by wearable sensors and on developing models and smart decision strategies to optimize the incorporation of these novel inputs into T1D interventions.
Keywords: big data; consumer health informatics; mobile health; survey; type 1 diabetes; wearable sensors.
©Elena Daskalaki, Anne Parkinson, Nicola Brew-Sam, Md Zakir Hossain, David O'Neal, Christopher J Nolan, Hanna Suominen. Originally published in the Journal of Medical Internet Research (https://www.jmir.org), 08.04.2022.
Conflict of interest statement
Conflicts of Interest: None declared.
Figures


Similar articles
-
Integrating Multiple Inputs Into an Artificial Pancreas System: Narrative Literature Review.JMIR Diabetes. 2022 Feb 24;7(1):e28861. doi: 10.2196/28861. JMIR Diabetes. 2022. PMID: 35200143 Free PMC article. Review.
-
Advanced Diabetes Management Using Artificial Intelligence and Continuous Glucose Monitoring Sensors.Sensors (Basel). 2020 Jul 10;20(14):3870. doi: 10.3390/s20143870. Sensors (Basel). 2020. PMID: 32664432 Free PMC article. Review.
-
Physiological Sensors Equipped in Wearable Devices for Management of Long COVID Persisting Symptoms: Scoping Review.J Med Internet Res. 2025 Mar 26;27:e69506. doi: 10.2196/69506. J Med Internet Res. 2025. PMID: 40137051 Free PMC article.
-
Type 1 Diabetes Hypoglycemia Prediction Algorithms: Systematic Review.JMIR Diabetes. 2022 Jul 21;7(3):e34699. doi: 10.2196/34699. JMIR Diabetes. 2022. PMID: 35862181 Free PMC article. Review.
-
Data-driven modeling and prediction of blood glucose dynamics: Machine learning applications in type 1 diabetes.Artif Intell Med. 2019 Jul;98:109-134. doi: 10.1016/j.artmed.2019.07.007. Epub 2019 Jul 26. Artif Intell Med. 2019. PMID: 31383477 Review.
Cited by
-
Variability of the Skin Temperature from Wrist-Worn Device for Definition of Novel Digital Biomarkers of Glycemia.Sensors (Basel). 2025 Jun 28;25(13):4038. doi: 10.3390/s25134038. Sensors (Basel). 2025. PMID: 40648295 Free PMC article.
-
Cardiovascular autonomic neuropathy in diabetes: an update with a focus on management.Diabetologia. 2024 Dec;67(12):2611-2625. doi: 10.1007/s00125-024-06242-0. Epub 2024 Aug 9. Diabetologia. 2024. PMID: 39120767 Free PMC article. Review.
-
Machine learning for detecting DNA attachment on SPR biosensor.Sci Rep. 2023 Mar 6;13(1):3742. doi: 10.1038/s41598-023-29395-1. Sci Rep. 2023. PMID: 36879019 Free PMC article.
-
Patent analysis of digital sensors for continuous glucose monitoring.Front Public Health. 2023 Aug 9;11:1205903. doi: 10.3389/fpubh.2023.1205903. eCollection 2023. Front Public Health. 2023. PMID: 37621612 Free PMC article. Review.
-
Digital Gaming and Exercise Among Youth With Type 1 Diabetes: Cross-Sectional Analysis of Data From the Type 1 Diabetes Exercise Initiative Pediatric Study.JMIR Pediatr Parent. 2024 Jun 13;7:e57198. doi: 10.2196/57198. JMIR Pediatr Parent. 2024. PMID: 38889077 Free PMC article.
References
-
- Atkinson MA, Eisenbarth GS, Michels AW. Type 1 diabetes. Lancet. 2014 Jan 04;383(9911):69–82. doi: 10.1016/S0140-6736(13)60591-7. http://europepmc.org/abstract/MED/23890997 S0140-6736(13)60591-7 - DOI - PMC - PubMed
-
- Mortensen HB, Robertson K, Aanstoot H, Danne T, Holl R, Hougaard P, Atchison J, Chiarelli F, Daneman D, Dinesen B, Dorchy H, Garandeau P, Greene S, Hoey H, Kaprio E, Kocova M, Martul P, Matsuura N, Schoenle E, Søvik O, Swift P, Tsou R, Vanelli M, Åman J. Insulin management and metabolic control of Type 1 diabetes mellitus in childhood and adolescence in 18 countries. Diabet Med. 1998 Sep;15(9):752–9. doi: 10.1002/(sici)1096-9136(199809)15:9<752::aid-dia678>3.0.co;2-w. doi: 10.1002/(sici)1096-9136(199809)15:9<752::aid-dia678>3.0.co;2-w. - DOI - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

