Donor leukocyte trafficking during human ex vivo lung perfusion
- PMID: 35396887
- PMCID: PMC9540615
- DOI: 10.1111/ctr.14670
Donor leukocyte trafficking during human ex vivo lung perfusion
Abstract
Background: Ex vivo lung perfusion (EVLP) is used to assess and preserve lungs prior to transplantation. However, its inherent immunomodulatory effects are not completely understood. We examine perfusate and tissue compartments to determine the change in immune cell composition in human lungs maintained on EVLP.
Methods: Six human lungs unsuitable for transplantation underwent EVLP. Tissue and perfusate samples were obtained during cold storage and at 1-, 3- and 6-h during perfusion. Flow cytometry, immunohistochemistry, and bead-based immunoassays were used to measure leukocyte composition and cytokines. Mean values between baseline and time points were compared by Student's t test.
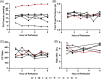
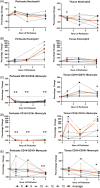
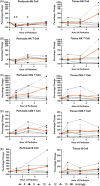
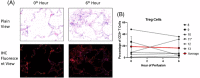
Results: During the 1st hour of perfusion, perfusate neutrophils increased (+22.2 ± 13.5%, p < 0.05), monocytes decreased (-77.5 ± 8.6%, p < 0.01) and NK cells decreased (-61.5 ± 22.6%, p < 0.01) compared to cold storage. In contrast, tissue neutrophils decreased (-22.1 ± 12.2%, p < 0.05) with no change in monocytes and NK cells. By 6 h, perfusate neutrophils, NK cells, and tissue neutrophils were similar to baseline. Perfusate monocytes remained decreased, while tissue monocytes remained unchanged. There was no significant change in B cells or T cell subsets. Pro-inflammatory cytokines (IL-1b, G-CSF, IFN-gamma, CXCL2, CXCL1 granzyme A, and granzyme B) and lymphocyte activating cytokines (IL-2, IL-4, IL-6, IL-8) increased during perfusion.
Conclusions: Early mobilization of innate immune cells occurs in both perfusate and tissue compartments during EVLP, with neutrophils and NK cells returning to baseline and monocytes remaining depleted after 6 h. The immunomodulatory effect of EVLP may provide a therapeutic window to decrease the immunogenicity of lungs prior to transplantation.
Keywords: donor leukocyte; ex vivo lung perfusion; immune modulation.
© 2022 The Authors. Clinical Transplantation published by John Wiley & Sons Ltd.
Conflict of interest statement
The authors have no conflict of interest.
Figures

Similar articles
-
Normothermic Ex Vivo Lung Perfusion in Brain-dead Donors Reduces Inflammatory Cytokines and Toll-like Receptor Expression.Iran J Allergy Asthma Immunol. 2016 Oct;15(5):340-354. Iran J Allergy Asthma Immunol. 2016. PMID: 27917620
-
Male versus female inflammatory response after brain death model followed by ex vivo lung perfusion.Biol Sex Differ. 2024 Jan 29;15(1):11. doi: 10.1186/s13293-024-00581-8. Biol Sex Differ. 2024. PMID: 38287395 Free PMC article.
-
Protein expression profiling predicts graft performance in clinical ex vivo lung perfusion.Ann Surg. 2015 Mar;261(3):591-7. doi: 10.1097/SLA.0000000000000974. Ann Surg. 2015. PMID: 25371129
-
The Conversional Efficacy of Ex Vivo Lung Perfusion and Clinical Outcomes in Patients Undergoing Transplantation of Donor Lungs by Ex Vivo Lung Perfusion: A Meta-Analysis.Ann Transplant. 2019 Dec 27;24:647-660. doi: 10.12659/AOT.919242. Ann Transplant. 2019. PMID: 31879416 Free PMC article.
-
Outcomes of marginal donors for lung transplantation after ex vivo lung perfusion: A systematic review and meta-analysis.J Thorac Cardiovasc Surg. 2020 Feb;159(2):720-730.e6. doi: 10.1016/j.jtcvs.2019.07.087. Epub 2019 Aug 25. J Thorac Cardiovasc Surg. 2020. PMID: 31548078
Cited by
-
Characterization and Proteomic Analyses of Proinflammatory Cytokines in a Mouse Model of Liver Transplant Rejection.Oxid Med Cell Longev. 2022 Aug 12;2022:5188584. doi: 10.1155/2022/5188584. eCollection 2022. Oxid Med Cell Longev. 2022. PMID: 35993024 Free PMC article.
-
The Potential Importance of CXCL1 in the Physiological State and in Noncancer Diseases of the Cardiovascular System, Respiratory System and Skin.Int J Mol Sci. 2022 Dec 22;24(1):205. doi: 10.3390/ijms24010205. Int J Mol Sci. 2022. PMID: 36613652 Free PMC article. Review.
References
-
- van Suylen V, Luijk B, Hoek RAS, et al. a multicenter study on long‐term outcomes after lung transplantation comparing donation after circulatory death and donation after brain death. Am J Transplant. 2017;17(10):2679–2686. - PubMed
-
- Chambers DC, Yusen RD, Cherikh WS, et al. The registry of the international society for heart and lung transplantation: thirty‐fourth adult lung and heart‐lung transplantation report‐2017; focus theme: allograft ischemic time. J Heart Lung Transplant. 2017;36(10):1047–1059. - PubMed
-
- Cypel M, Liu M, Rubacha M, et al. Functional repair of human donor lungs by IL‐10 gene therapy. Sci Transl Med. 2009;1(4):4ra9. - PubMed
-
- Nakajima D, Chen F, Yamada T, et al. Reconditioning of lungs donated after circulatory death with normothermic ex vivo lung perfusion. J Heart Lung Transplant. 2012;31(2):187–193. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

