Long noncoding RNA ANRIL up-regulates CCND1 via sponging miR-98-5p to promote TGF-β1-induced human airway smooth muscle cell proliferation, migration, and extracellular matrix deposition
- PMID: 35396910
- PMCID: PMC11896278
- DOI: 10.1002/kjm2.12538
Long noncoding RNA ANRIL up-regulates CCND1 via sponging miR-98-5p to promote TGF-β1-induced human airway smooth muscle cell proliferation, migration, and extracellular matrix deposition
Abstract
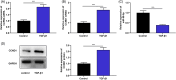
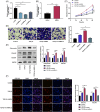
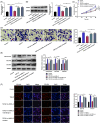
Excessive proliferation and migration of airway smooth muscle cell (ASMC) contribute to asthma pathogenesis. Long noncoding RNAs (lncRNAs) are reported to take part in asthma pathogenesis. This study is targeted at deciphering the role of the lncRNA antisense noncoding RNA in the INK4 locus (ANRIL) in ASMC proliferation, migration and extracellular matrix (ECM) deposition. qRT-PCR was performed to determine ANRIL, miR-98-5p, and cyclin D1 (CCND1) mRNA expression levels in transforming growth factor-β1 (TGF-β1)-treated ASMCs. CCK-8 and Transwell assays were employed to examine ASMC proliferation and migration, respectively. Dual-luciferase reporter gene assay and RNA immunoprecipitation assay were carried out for analyzing the targeted relationship of miR-98-5p with ANRIL or CCND1 mRNA 3'-UTR. The levels of CCND1 and ECM proteins (such as fibronectin, COL3A1, and COL1A2) in ASMCs were detected through Western blot. In this work, we found that ANRIL and CCND1 were up-regulated in TGF-β1-treated ASMCs, whereas miR-98-5p was down-regulated. ANRIL overexpression facilitated the proliferation, ECM deposition and migration of TGF-β1-induced ASMCs, while knocking down ANRIL had the opposite effect. Furthermore, ANRIL targeted miR-98-5p directly, and CCND1 was miR-98-5p's downstream target. ANRIL indirectly increased CCND1 expression in ASMCs via competitively binding to miR-98-5p. MiR-98-5p inhibition or CCND1 overexpression counteracted the inhibiting effect that ANRIL knockdown had on TGF-β1-stimulated ASMC proliferation, migration and ECM deposition. In conclusion, ANRIL indirectly up-regulates CCND1 expression by targeting miR-98-5p to promote ASMC proliferation, migration and ECM deposition, thus facilitating the pathogenesis of asthma.
Keywords: ANRIL; CCND1; MiR-98-5p; airway smooth muscle cell; asthma.
© 2022 The Authors. The Kaohsiung Journal of Medical Sciences published by John Wiley & Sons Australia, Ltd on behalf of Kaohsiung Medical University.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
-
- Pavord ID, Beasley R, Agusti A, Anderson GP, Bel E, Brusselle G, et al. After asthma: redefining airways diseases. Lancet. 2018;391(10118):350–400. - PubMed
-
- Fang L, Roth M, S'ng CT, Tamm M, Han B, Hoang BX. Zinc salicylate reduces airway smooth muscle cells remodelling by blocking mTOR and activating p21(Waf1/Cip1). J Nutr Biochem. 2021;89:108563. - PubMed
-
- Manuyakorn W, Kamchaisatian W, Atamasirikul K, Sasisakulporn C, Direkwattanachai C, Benjaponpitak S. Serum TGF‐beta1 in atopic asthma. Asian Pac J Allergy Immunol. 2008;26(4):185–9. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

