Rethinking Mitchell's Chemiosmotic Theory: Potassium Dominates Over Proton Flux to Drive Mitochondrial F1Fo-ATP Synthase
- PMID: 35399493
- PMCID: PMC8991028
- DOI: 10.1093/function/zqac012
Rethinking Mitchell's Chemiosmotic Theory: Potassium Dominates Over Proton Flux to Drive Mitochondrial F1Fo-ATP Synthase
Figures
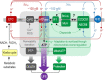
Comment on
-
ATP Synthase K+- and H+-fluxes Drive ATP Synthesis and Enable Mitochondrial K+-"Uniporter" Function: II. Ion and ATP Synthase Flux Regulation.Function (Oxf). 2022 Jan 27;3(2):zqac001. doi: 10.1093/function/zqac001. eCollection 2022. Function (Oxf). 2022. PMID: 35187492 Free PMC article.
-
ATP Synthase K+- and H+-Fluxes Drive ATP Synthesis and Enable Mitochondrial K+-"Uniporter" Function: I. Characterization of Ion Fluxes.Function (Oxf). 2021 Dec 13;3(2):zqab065. doi: 10.1093/function/zqab065. eCollection 2022. Function (Oxf). 2021. PMID: 35229078 Free PMC article.
References
-
- Mitchell P. Coupling of phosphorylation to electron and hydrogen transfer by a chemi-osmotic type of mechanism. Nature. 1961;191(4784):144–148. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
