Monoallelic Heb/Tcf12 Deletion Reduces the Requirement for NOTCH1 Hyperactivation in T-Cell Acute Lymphoblastic Leukemia
- PMID: 35401501
- PMCID: PMC8987207
- DOI: 10.3389/fimmu.2022.867443
Monoallelic Heb/Tcf12 Deletion Reduces the Requirement for NOTCH1 Hyperactivation in T-Cell Acute Lymphoblastic Leukemia
Abstract
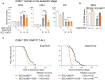
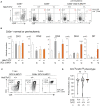
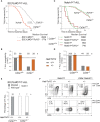
Early T-cell development is precisely controlled by E proteins, that indistinguishably include HEB/TCF12 and E2A/TCF3 transcription factors, together with NOTCH1 and pre-T cell receptor (TCR) signalling. Importantly, perturbations of early T-cell regulatory networks are implicated in leukemogenesis. NOTCH1 gain of function mutations invariably lead to T-cell acute lymphoblastic leukemia (T-ALL), whereas inhibition of E proteins accelerates leukemogenesis. Thus, NOTCH1, pre-TCR, E2A and HEB functions are intertwined, but how these pathways contribute individually or synergistically to leukemogenesis remain to be documented. To directly address these questions, we leveraged Cd3e-deficient mice in which pre-TCR signaling and progression through β-selection is abrogated to dissect and decouple the roles of pre-TCR, NOTCH1, E2A and HEB in SCL/TAL1-induced T-ALL, via the use of Notch1 gain of function transgenic (Notch1ICtg) and Tcf12+/- or Tcf3+/- heterozygote mice. As a result, we now provide evidence that both HEB and E2A restrain cell proliferation at the β-selection checkpoint while the clonal expansion of SCL-LMO1-induced pre-leukemic stem cells in T-ALL is uniquely dependent on Tcf12 gene dosage. At the molecular level, HEB protein levels are decreased via proteasomal degradation at the leukemic stage, pointing to a reversible loss of function mechanism. Moreover, in SCL-LMO1-induced T-ALL, loss of one Tcf12 allele is sufficient to bypass pre-TCR signaling which is required for Notch1 gain of function mutations and for progression to T-ALL. In contrast, Tcf12 monoallelic deletion does not accelerate Notch1IC-induced T-ALL, indicating that Tcf12 and Notch1 operate in the same pathway. Finally, we identify a tumor suppressor gene set downstream of HEB, exhibiting significantly lower expression levels in pediatric T-ALL compared to B-ALL and brain cancer samples, the three most frequent pediatric cancers. In summary, our results indicate a tumor suppressor function of HEB/TCF12 in T-ALL to mitigate cell proliferation controlled by NOTCH1 in pre-leukemic stem cells and prevent NOTCH1-driven progression to T-ALL.
Keywords: E2A/TCF3; HEB/TCF12; LMO1; NOTCH1; SCL/TAL1; T-cell acute lymphoblastic leukemia; tumor suppressor genes.
Copyright © 2022 Veiga, Tremblay, Gerby, Herblot, Haman, Gendron, Lemieux, Zúñiga-Pflücker, Hébert, Cohen and Hoang.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures



Similar articles
-
Modeling T-cell acute lymphoblastic leukemia induced by the SCL and LMO1 oncogenes.Genes Dev. 2010 Jun 1;24(11):1093-105. doi: 10.1101/gad.1897910. Genes Dev. 2010. PMID: 20516195 Free PMC article.
-
SCL, LMO1 and Notch1 reprogram thymocytes into self-renewing cells.PLoS Genet. 2014 Dec 18;10(12):e1004768. doi: 10.1371/journal.pgen.1004768. eCollection 2014 Dec. PLoS Genet. 2014. PMID: 25522233 Free PMC article.
-
The TAL1 complex targets the FBXW7 tumor suppressor by activating miR-223 in human T cell acute lymphoblastic leukemia.J Exp Med. 2013 Jul 29;210(8):1545-57. doi: 10.1084/jem.20122516. Epub 2013 Jul 15. J Exp Med. 2013. PMID: 23857984 Free PMC article.
-
SCL/TAL1 in Hematopoiesis and Cellular Reprogramming.Curr Top Dev Biol. 2016;118:163-204. doi: 10.1016/bs.ctdb.2016.01.004. Epub 2016 Feb 18. Curr Top Dev Biol. 2016. PMID: 27137657 Review.
-
E Protein Transcription Factors as Suppressors of T Lymphocyte Acute Lymphoblastic Leukemia.Front Immunol. 2022 Apr 20;13:885144. doi: 10.3389/fimmu.2022.885144. eCollection 2022. Front Immunol. 2022. PMID: 35514954 Free PMC article. Review.
Cited by
-
Regulation of the Signal-Dependent E Protein HEBAlt Through a YYY Motif Is Required for Progression Through T Cell Development.Front Immunol. 2022 Aug 3;13:848577. doi: 10.3389/fimmu.2022.848577. eCollection 2022. Front Immunol. 2022. PMID: 35990644 Free PMC article.
-
IL-15 Prevents the Development of T-ALL from Aberrant Thymocytes with Impaired DNA Repair Functions and Increased NOTCH1 Activation.Cancers (Basel). 2023 Jan 21;15(3):671. doi: 10.3390/cancers15030671. Cancers (Basel). 2023. PMID: 36765626 Free PMC article.
-
Nuclear Tubulin Enhances CXCR4 Transcription and Promotes Chemotaxis Through TCF12 Transcription Factor in human Hematopoietic Stem Cells.Stem Cell Rev Rep. 2023 Jul;19(5):1328-1339. doi: 10.1007/s12015-023-10543-z. Epub 2023 Apr 17. Stem Cell Rev Rep. 2023. PMID: 37067645
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

