Plasmodium sporozoite disintegration during skin passage limits malaria parasite transmission
- PMID: 35403820
- PMCID: PMC9253755
- DOI: 10.15252/embr.202254719
Plasmodium sporozoite disintegration during skin passage limits malaria parasite transmission
Abstract
During transmission of malaria-causing parasites from mosquitoes to mammals, Plasmodium sporozoites migrate rapidly in the skin to search for a blood vessel. The high migratory speed and narrow passages taken by the parasites suggest considerable strain on the sporozoites to maintain their shape. Here, we show that the membrane-associated protein, concavin, is important for the maintenance of the Plasmodium sporozoite shape inside salivary glands of mosquitoes and during migration in the skin. Concavin-GFP localizes at the cytoplasmic periphery and concavin(-) sporozoites progressively round up upon entry of salivary glands. Rounded concavin(-) sporozoites fail to pass through the narrow salivary ducts and are rarely ejected by mosquitoes, while normally shaped concavin(-) sporozoites are transmitted. Strikingly, motile concavin(-) sporozoites disintegrate while migrating through the skin leading to parasite arrest or death and decreased transmission efficiency. Collectively, we suggest that concavin contributes to cell shape maintenance by riveting the plasma membrane to the subtending inner membrane complex. Interfering with cell shape maintenance pathways might hence provide a new strategy to prevent a malaria infection.
Keywords: cell migration; cell shape maintenance; gliding motility; malaria transmission; pellicle.
© 2022 The Authors. Published under the terms of the CC BY NC ND 4.0 license.
Figures

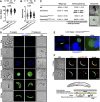
Blood‐stage growth rate of concavin(−) parasites in comparison to wild‐type. Data points represent parasites growing in individual mice, and n indicates total number of mice. P‐value calculated using the Mann–Whitney test. Shown is the mean ± SEM.
Gametocyte to ookinete conversion of three biological replicates. P‐value calculated using the Mann–Whitney test. Shown is the mean ± SEM.
Average speed of moving ookinetes. Data points represent individual ookinetes, and n indicates total number of cells from three biological replicates. P‐value calculated using the Kruskal–Wallis test followed by Dunns multiple comparison.
Ookinete images of wild‐type and concavin(−) parasites revealing similar cell shapes. Scale bar 5 µm.
Oocyst development of concavin(−) parasites compared to wild‐type. Data points represent individual midguts observed between d12‐17 post infection from 3 independent cage feeds. Shown is the mean ± SEM. P‐value calculated using the Kruskal–Wallis test followed by Dunns multiple comparison.
Mosquito infections resulted in deformed sporozoites in the salivary gland. Shown are example images of different sporozoites arranged to illustrate their rounding up over time.
Quantification of sporozoite rounding up over time in the midgut (MG) the haemolymph (HL) and salivary gland (SG) at the indicated days post infection from three biological replicates. Sporozoites were classified as either normal, deformed or round as illustrated in F.
Average speed of salivary gland sporozoites. Data points represent individual sporozoites, and n indicates total number from three biological replicates. P‐values calculated using the Kruskal–Wallis test followed by the Dunns multiple comparison test.
Selected trajectories of manually tracked sporozoites.

Oocyst development of wild‐type and concavin(−) parasites in A. gambiae mosquitoes. Data points represent individual midguts observed between d12‐17 post infection (left). Shown is the mean ± SEM. P‐value calculated using the Mann–Whitney test. Quantification of sporozoite forms in the midgut and the salivary gland at indicated time points (right).
Quantification of sporozoite forms in the midgut, the haemolymph and the salivary gland at indicated time points in A. stephensi infections.
- A
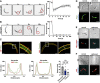
Oocyst development of concavin(−) parasites complemented with either the Plasmodium berghei gene or the P. falciparum orthologue fused to GFP. Data points represent individual midguts observed between d12‐17 post infection of three independent cage feeds. Shown is the mean ± SEM. P‐values are calculated using the Kruskal–Wallis test followed by the Dunns multiple comparison test.
- B
Average speed of salivary gland sporozoites. Data points represent individual sporozoites of three independent biological replicates. Shown is the mean ± SEM. P‐values are calculated using the Kruskal–Wallis test followed by the Dunns multiple comparison test.
- C
Sum table of mosquito infections of wild‐type, concavin(−) and complemented lines expressing either P. berghei (Pb) or P. falciparum (Pf) concavin‐GFP. Numbers determined on d17 post mosquito infection. Infection rate contains three different countings of at least 20 mosquitoes from different mosquito infections. Note the difference of normally shaped and total sporozoites for concavin(−) parasites. Images show infection of salivary glands. Scale bars: 100 µm.
- D
Pb concavin‐GFP and Pf concavin‐GFP localization in blood and mosquito stages. Nuclei (blue) stained with Hoechst. Scale bar: 5 µm.
- E
Localization of P. berghei concavin‐GFP in liver stages. Nuclei (blue) stained with Hoechst. Scale bar: 5 µm.
- F
Immunofluorescence images of permeabilized and unpermeabilized concavin‐GFP expressing salivary gland sporozoites stained with an anti‐GFP antibody. Note that a GFP signal could only be detected after permeabilization, excluding concavin localization on the parasite surface as illustrated in the model. Scale bar: 5 µm.
- A, B
Time series of concavin‐GFP (A) PhiL1‐GFP (B) sporozoites before and after bleaching and quantification of the fluorescence signal over time of three technical replicates. * indicates time of bleaching. Shown is the mean ± SD.
- C
Super resolution (STED) imaging of PhiL1‐GFP and CSP as well as concavin‐GFP and CSP. Cells were stained with an anti‐GFP antibody in combination with Atto‐594 (green) in addition to an anti‐CSP staining in combination with Atto‐647 (red). Images were deconvolved using the Richardson‐Lucy algorithm. The distance between the 2 signal peaks was measured using the plot profile of the respective channels in Fiji. Data points represent distance in individual sporozoites at the center of the cell from two independent biological replicates. P‐values are calculated using the Kruskal Wallis test followed by the Dunns multiple comparison test. Scale bar: 1 µm.
- D
Localization of PhiL1‐GFP (green) in normal and deformed concavin(−) parasites. Nuclei (blue) stained with Hoechst. Scale bar 5 µm.
- E
Localization of SiR‐Tubulin (red) in concavin(−) parasites. Nuclei (blue) stained with Hoechst. Scale bar 5 µm.
- F
Localization of CSP (red) in concavin(−) parasites. Nuclei (blue) stained with Hoechst. Scale bar 5 µm.


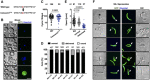
Transmission electron micrographs of wild‐type sporozoites with highlighted secretory organelles in magenta, IMC in yellow, microtubules in green and plasma membrane in blue. Scale bar 500 nm.
Transmission electron microscopy of concavin(−) sporozoites with highlighted secretory organelles as in A; rhoptries in red and nucleus in turquoise. Scale bars: 500 nm.
Reconstruction of serial sections of a complete rounded up concavin(−) sporozoite; plasma membrane in blue and IMC in yellow. Scale bar 1 µm. In total, 6 parasites were reconstructed from two different preparations.

Transmission electron microscopy images of different concavin(−) sporozoites. Scale bars: 500 nm.
Scanning electron microscopy images from serial sections of a concavin(−) sporozoites. Scale bar: 500 nm.
Cysteine 7 is predicted to be palmitoylated and was changed into alanine in the concavinC7A‐GFP mutant.
Expression and localization of concavinC7A‐GFP in blood stage parasites. Scale bar 5 µm.
Oocysts number in infected mosquitoes. Data points represent individual midguts observed between d12‐17 post infection of three independent cage feeds. Shown is the mean ± SEM. P‐value calculated using the Mann Whitney test.
Quantification of concavinC7A‐GFP cell shapes at the indicated days from midgut (MG) or salivary gland (SG) derived sporozoites. Numbers above bars indicate investigated sporozoites.
Average speed of salivary gland sporozoites. Data points represent individual sporozoites from three independent biological replicates. Shown is the mean ± SEM. P‐values are calculated using the Kruskal Wallis test followed by the Dunns multiple comparison test.
Localization of concavinC7A‐GFP in normal and deformed sporozoites (arrowheads point to deformations). Scale bar: 5 µm.
- A
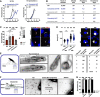
Growth curve of blood stage parasites in C57BL/6 mice infected by the bite of 10 mosquitoes (left) or 10,000 sporozoites intra venously (right) at two different times post mosquito infection as indicated. Shown is the mean ± SEM. Mice infected with concavin(−) parasites by bite represent two independent biological replicates with three mice each. Wild‐type bite back and concavin(−) intra venous injections represent one biological replicate using 3 or 4 mice respectively.
- B
Sum table of infected mice from A with pre‐patency period (time to detect a blood stage infection) and parasitemia on day 6. Note that in concavin(−) by bite infected mice not all develop a blood stage infection.
- C
Liver cell invasion assay of concavin‐gfp (WT) and concavin(−) parasites. Parasites are positively stained for CSP in case they remain extracellular. Both, normal and deformed parasites were detected intracellularly. Graph shows quantification of CSP positive (red, hence extracellular) and negative (grey, hence intracellular) sporozoites. Small graph shows the percentage of deformed and normally shaped intracellular concavin(−) sporozoites. Data points represent the 4 individual biological replicates with n indicating the numbers of sporozoites observed. Shown is the mean ± SEM. P‐values are calculated using the Mann–Whitney test. Scale bar: 5 µm.
- D
Liver‐stage development of concavin(−) parasites compared to wild‐type, both expressing cytoplasmic GFP (white). Parasite size was measured 24 and 48 h post infection. Data points represent individual parasites from four independent biological replicates. Shown is the mean ± SEM. P‐values calculated using the Mann Whitney test. Scale bar: 5 µm.
- E
Sporozoite ejection of immobilized concavin(−) infected mosquitoes on glass slides and quantification of ejected sporozoites from 20 mosquitoes. * indicates individual sporozoites in the ejected saliva. Scale bar: 10 µm.
- F, G
Only normally shaped concavin(−) sporozoites released by salivary glands move on helical paths (arrows) through polyacrylamide gels that mimic the skin (F). Scale bar: 10 µm. (G): Quantification of two individual experiments; only normal shaped sporozoites were able to migrate through the gel.
- A
Percentage of mosquitoes depositing WT or concavin(−) sporozoites in the skin of a mouse during a bite (left) and sporozoites deposited during a mosquito bite (right). P‐values are calculated using the Fishers exact test (left) and the Mann Whitney test (right). Bite transmission experiments were repeated over three independent mosquito infections for the KO and two independent mosquito infections for the WT. For each mosquito infection 2–3 mice were imaged, for a total of 3–5 ears.
- B
Maximum fluorescence intensity projections encoded by color for time from movies showing migrating sporozoites after mosquito‐bite transmission. Graphs and camembert diagrams below show fraction of motile and immotile sporozoites after 10, 20 and 30 min of recording, numbers analysed as well as sporozoite speed. Pooled data from 4–5 WT or 5–8 concavin(−) bite sites per time point. Scale bar: 50 µm. P‐values are calculated using the Mann Whitney test.
- C
Percentage of motile WT or concavin(−) sporozoites entering blood vessels during the first hour following micro‐injection. Number above bars shows numbers of sporozoites observed. Pooled data from four WT or five concavin(−) experiments. P‐values are calculated using the Fishers exact test.
- D, E
(D) Deformation and (E) disintegration of concavin(−) sporozoites migrating in the skin. Individual images corresponding to the frames shown on the right are indicated in distinct colours in the maximum projection (left) of 13‐min movies. Arrowheads indicate constrictions of the parasites. Graphs below time‐lapse show that deformation and disintegration are preceded by a decrease in speed. Color of dots correspond to the time‐points displayed in the time‐lapse images. Scale bars: 10 µm.
- F
Percentage of deformation and disintegration events observed in 54 WT and 57 concavin(−) sporozoites that were tracked for at least 10 min. Pooled data from 5 WT or 10 concavin(−) bite sites.
- G
Consequence of deformation and disintegration from the 22 sporozoites in F include immobilization and parasite death.
References
-
- Aliprandini E, Tavares J, Panatieri RH, Thiberge S, Yamamoto MM, Silvie O, Ishino T, Yuda M, Dartevelle S, Traincard F et al (2018) Cytotoxic anti‐circumsporozoite antibodies target malaria sporozoites in the host skin. Nat Microbiol 3: 1224–1233 - PubMed
-
- Amino R, Giovannini D, Thiberge S, Gueirard P, Boisson B, Dubremetz JF, Prévost MC, Ishino T, Yuda M, Ménard R (2008) Host cell traversal is important for progression of the malaria parasite through the dermis to the liver. Cell Host Microbe 3: 88–96 - PubMed
-
- Amino R, Thiberge S, Martin B, Celli S, Shorte S, Frischknecht F, Ménard R (2006) Quantitative imaging of Plasmodium transmission from mosquito to mammal. Nat Med 12: 220–224 - PubMed
-
- Amino R, Thiberge S, Blazquez S, Baldacci P, Renaud O, Shorte S, Ménard R (2007) Imaging malaria sporozoites in the dermis of the mammalian host. Nat Protoc 2: 1705–1712 - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

