Validation-based model selection for 13C metabolic flux analysis with uncertain measurement errors
- PMID: 35404953
- PMCID: PMC9022838
- DOI: 10.1371/journal.pcbi.1009999
Validation-based model selection for 13C metabolic flux analysis with uncertain measurement errors
Abstract
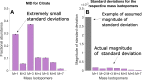
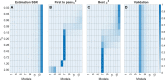
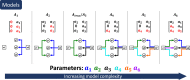
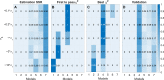
Accurate measurements of metabolic fluxes in living cells are central to metabolism research and metabolic engineering. The gold standard method is model-based metabolic flux analysis (MFA), where fluxes are estimated indirectly from mass isotopomer data with the use of a mathematical model of the metabolic network. A critical step in MFA is model selection: choosing what compartments, metabolites, and reactions to include in the metabolic network model. Model selection is often done informally during the modelling process, based on the same data that is used for model fitting (estimation data). This can lead to either overly complex models (overfitting) or too simple ones (underfitting), in both cases resulting in poor flux estimates. Here, we propose a method for model selection based on independent validation data. We demonstrate in simulation studies that this method consistently chooses the correct model in a way that is independent on errors in measurement uncertainty. This independence is beneficial, since estimating the true magnitude of these errors can be difficult. In contrast, commonly used model selection methods based on the χ2-test choose different model structures depending on the believed measurement uncertainty; this can lead to errors in flux estimates, especially when the magnitude of the error is substantially off. We present a new approach for quantification of prediction uncertainty of mass isotopomer distributions in other labelling experiments, to check for problems with too much or too little novelty in the validation data. Finally, in an isotope tracing study on human mammary epithelial cells, the validation-based model selection method identified pyruvate carboxylase as a key model component. Our results argue that validation-based model selection should be an integral part of MFA model development.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures








References
-
- Berg JM, Tymoczko JL, Stryer L. Biochemistry. [Internet]. 7th ed. W.H. Freeman & Company; 2010; 2010. Available from: https://login.e.bibl.liu.se/login?url = https://search.ebscohost.com/login.aspx?direct=true&AuthType=ip,uid&db=c...

