Phenotypical Screening on Neuronal Plasticity in Hippocampal-Prefrontal Cortex Connectivity Reveals an Antipsychotic with a Novel Profile
- PMID: 35406745
- PMCID: PMC8997950
- DOI: 10.3390/cells11071181
Phenotypical Screening on Neuronal Plasticity in Hippocampal-Prefrontal Cortex Connectivity Reveals an Antipsychotic with a Novel Profile
Abstract
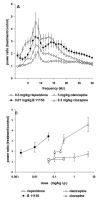
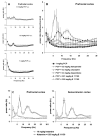
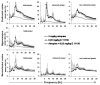
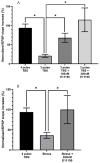
Dysfunction in the hippocampus-prefrontal cortex (H-PFC) circuit is a critical determinant of schizophrenia. Screening of pyridazinone-risperidone hybrids on this circuit revealed EGIS 11150 (S 36549). EGIS 11150 induced theta rhythm in hippocampal slice preparations in the stratum lacunosum molecular area of CA1, which was resistant to atropine and prazosin. EGIS 11150 enhanced H-PFC coherence, and increased the 8−9 Hz theta band of the EEG power spectrum (from 0.002 mg/kg i.p, at >30× lower doses than clozapine, and >100× for olanzapine, risperidone, or haloperidol). EGIS 11150 fully blocked the effects of phencyclidine (PCP) or ketamine on EEG. Inhibition of long-term potentiation (LTP) in H-PFC was blocked by platform stress, but was fully restored by EGIS 11150 (0.01 mg/kg i.p.), whereas clozapine (0.3 mg/kg ip) only partially restored LTP. EGIS 11150 has a unique electrophysiological profile, so phenotypical screening on H-PFC connectivity can reveal novel antipsychotics.
Keywords: EGIS 11150; S 36549; dysconnectivity; ketamine; phencyclidine; schizophrenia; theta rhythm.
Conflict of interest statement
M. Spedding was an employee of Servier (cosponsor/discoverer/developer of EGIS 11150), E. Schenker an employee of Servier. G. Szénási, G. Lévay, J. Barkóczy, L. G. Hársing Jr., I. Gacsályi are/were (at the time of studies) employees of EGIS (cosponsor/discoverer/developper of EGIS 11150). C. Sebban, T.M. Jay, C. Rocher, B. Tesolin-Decros, I. Thomson, M. Cunningham, M.A. Whittington, L.-A. Etherington, J. Lambert performed contracted studies on EGIS 11150.
Figures




References
-
- Carter C.S., Barch D.M., Buchanan R.W., Bullmore E., Krystal J.H., Cohen J., Geyer M., Green M., Nuechterlein K.H., Robbins T., et al. Identifying Cognitive Mechanisms Targeted for Treatment Development in Schizophrenia: An Overview of the First Meeting of the Cognitive Neuroscience Treatment Research to Improve Cognition in Schizophrenia Initiative. Biol. Psychiatry. 2008;64:4–10. doi: 10.1016/j.biopsych.2008.03.020. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

