The Healing Capability of Clove Flower Extract (CFE) in Streptozotocin-Induced (STZ-Induced) Diabetic Rat Wounds Infected with Multidrug Resistant Bacteria
- PMID: 35408668
- PMCID: PMC9000752
- DOI: 10.3390/molecules27072270
The Healing Capability of Clove Flower Extract (CFE) in Streptozotocin-Induced (STZ-Induced) Diabetic Rat Wounds Infected with Multidrug Resistant Bacteria
Abstract
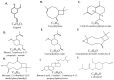
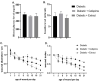
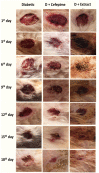
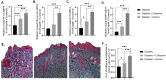
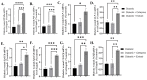
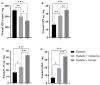
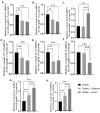
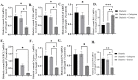
Treatment of diabetic foot ulcer (DFU) is of great challenge as it is shown to be infected by multidrug resistant bacteria (MDR bacteria). Sixty four bacterial isolates were isolated from DFU cases; antibiotic susceptibility tests were carried out for all of them. One bacterial isolate (number 11) was shown to resist the action of 8 out of 12 antibiotics used and was identified by both a Vitek-2 system and 16S rRNA fingerprints as belonging to Proteus mirabilis, and was designated Proteus mirabilis LC587231 (P. mirabilis). Clove flower extract (CFE) inhibited distinctively the P. mirabilis bacterium obtained. GC-MS spectroscopy showed that this CFE contained nine bioactive compounds. The effect of CFE on wound healing of Type 1 diabetic albino rats (Rattus norvegicus) was studied. The results indicated that topical application of CFE hydrogel improved wound size, wound index, mRNA expression of the wound healing markers (Coli1, MMP9, Fibronectin, PCNA, and TGFβ), growth factor signaling pathways (PPAR-α, PGC1-α, GLP-1, GLPr-1, EGF-β, EGF-βr, VEGF-β, and FGF-β), inflammatory cytokine expression (IL8, TNFα, NFKβ, IL1β, and MCP1), as well as anti-inflammatory cytokines (IL4 & IL10), pro-apoptotic markers (FAS, FAS-L, BAX, BAX/BCL-2, Caspase-3, P53, P38), as well as an antiapoptotic one (BCL2). Furthermore, it improved the wound oxidative state and reduced the wound microbial load, as the cefepime therapy improved the wound healing parameters. Based on the previous notions, it could be concluded that CFE represents a valid antibiotics alternative for DFU therapy since it improves diabetic wound healing and exerts antibacterial activity either in vitro or in vivo.
Keywords: MDR-Proteus mirabilis; Syzygium aromaticum; diabetic foot ulcer; growth factor; inflammatory markers.
Conflict of interest statement
The authors declare that they have no competing interest; financial or otherwise.
Figures

References
-
- Core D., Ahn J., Robert B.S., Lewis B., Katherine M.D., Raspovic M., Trapper D.P.M., Lalli A.J., Dane K., Wukich M.D. The Evaluation and Treatment of Diabetic Foot Ulcers and Diabetic Foot Infections. Foot Ankle Orthop. 2018;3:247301141878886. doi: 10.1177/2473011418788864. - DOI
-
- Enan G. Control of the regrowing bacteriocin resistance variants of Listeria monocytogenes LMG 10470 in Vitro and in Food by nisin-plantaricin UG1 mixture. Biotechnology. 2006;5:143–147. doi: 10.3923/biotech.2006.143.147. - DOI
-
- Abdel-Shafi S., Ouda S.M., Elbalat I., Enan G. Characterization and identification of multidrug resistant bacteria from some Egyptian patients. Biotechnology. 2013;12:65–73. doi: 10.3923/biotech.2013.65.73. - DOI
-
- Enan G., Abdel-Shafi S., Ouda S.M., El-Balat I. Genetic linkage of the antibiotic resistance ability in the Escherichia coli UR4 strain isolated from urine. J. Med Sci. 2013;13:261–268. doi: 10.3923/jms.2013.261.268. - DOI
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

