Skin Microbiota in Atopic Dermatitis
- PMID: 35408862
- PMCID: PMC8998607
- DOI: 10.3390/ijms23073503
Skin Microbiota in Atopic Dermatitis
Abstract
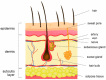
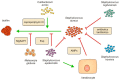
The skin microbiota represents an ecosystem composed of numerous microbial species interacting with each other, as well as with host epithelial and immune cells. The microbiota provides health benefits to the host by supporting essential functions of the skin and inhibiting colonization with pathogens. However, the disturbance of the microbial balance can result in dysbiosis and promote skin diseases, such as atopic dermatitis (AD). This review provides a current overview of the skin microbiota involvement in AD and its complex interplay with host immune response mechanisms, as well as novel therapeutic strategies for treating AD focused on restoring skin microbial homeostasis.
Keywords: atopic dermatitis; dysbiosis; skin microbiota.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources

