Distinctive Toll-like Receptors Gene Expression and Glial Response in Different Brain Regions of Natural Scrapie
- PMID: 35408945
- PMCID: PMC8998348
- DOI: 10.3390/ijms23073579
Distinctive Toll-like Receptors Gene Expression and Glial Response in Different Brain Regions of Natural Scrapie
Abstract
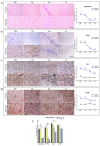
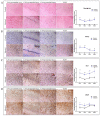
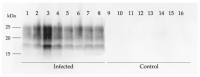
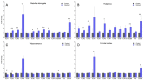
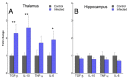
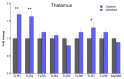
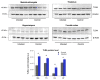
Prion diseases are chronic and fatal neurodegenerative diseases characterized by the accumulation of disease-specific prion protein (PrPSc), spongiform changes, neuronal loss, and gliosis. Growing evidence shows that the neuroinflammatory response is a key component of prion diseases and contributes to neurodegeneration. Toll-like receptors (TLRs) have been proposed as important mediators of innate immune responses triggered in the central nervous system in other human neurodegenerative diseases, including Alzheimer's disease, Parkinson's disease, and amyotrophic lateral sclerosis. However, little is known about the role of TLRs in prion diseases, and their involvement in the neuropathology of natural scrapie has not been studied. We assessed the gene expression of ovine TLRs in four anatomically distinct brain regions in natural scrapie-infected sheep and evaluated the possible correlations between gene expression and the pathological hallmarks of prion disease. We observed significant changes in TLR expression in scrapie-infected sheep that correlate with the degree of spongiosis, PrPSc deposition, and gliosis in each of the regions studied. Remarkably, TLR4 was the only gene upregulated in all regions, regardless of the severity of neuropathology. In the hippocampus, we observed milder neuropathology associated with a distinct TLR gene expression profile and the presence of a peculiar microglial morphology, called rod microglia, described here for the first time in the brain of scrapie-infected sheep. The concurrence of these features suggests partial neuroprotection of the hippocampus. Finally, a comparison of the findings in naturallyinfected sheep versus an ovinized mouse model (tg338 mice) revealed distinct patterns of TLRgene expression.
Keywords: astrocytes; microglia; neuroinflammation; prion; scrapie; toll-like receptors.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results. The pictures and figures in this manuscript are original data.
Figures


References
-
- Andreoletti O., Berthon P., Marc D., Sarradin P., Grosclaude J., van Keulen L., Schelcher F., Elsen J.M., Lantier F. Early accumulation of PrP(Sc) in gut-associated lymphoid and nervous tissues of susceptible sheep from a Romanov flock with natural scrapie. Pt 12J. Gen. Virol. 2000;81:3115–3126. doi: 10.1099/0022-1317-81-12-3115. - DOI - PubMed
-
- McBride P.A., Schulz-Schaeffer W.J., Donaldson M., Bruce M., Diringer H., Kretzschmar H.A., Beekes M. Early spread of scrapie from the gastrointestinal tract to the central nervous system involves autonomic fibers of the splanchnic and vagus nerves. J. Virol. 2001;75:9320–9327. doi: 10.1128/JVI.75.19.9320-9327.2001. - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

