Exploring the Effect of Methyl Jasmonate on the Expression of microRNAs Involved in Biosynthesis of Active Compounds of Rosemary Cell Suspension Cultures through RNA-Sequencing
- PMID: 35409063
- PMCID: PMC8998883
- DOI: 10.3390/ijms23073704
Exploring the Effect of Methyl Jasmonate on the Expression of microRNAs Involved in Biosynthesis of Active Compounds of Rosemary Cell Suspension Cultures through RNA-Sequencing
Abstract
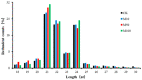
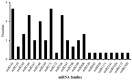
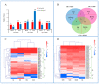
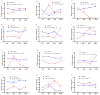
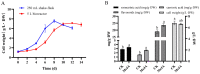
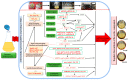
Our aim in the experiment was to study the effects of methyl jasmonates (MeJA) on the active compounds of rosemary suspension cells, the metabolites' change of contents under different concentrations of MeJA, including 0 (CK), 10 (M10), 50 (M50) and 100 μM MeJA (M100). The results demonstrated that MeJA treatments promoted the accumulation of rosmarinic acid (RA), carnosic acid (CA), flavonoids, jasmonate (JA), gibberellin (GA), and auxin (IAA); but reduced the accumulations of abscisic acid (ABA), salicylic acid (SA), and aspartate (Asp). In addition, 50 and 100 μM MeJA promoted the accumulation of alanine (Ala) and glutamate (Glu), and 50 μM MeJA promoted the accumulation of linoleic acid and alpha-linolenic acid in rosemary suspension cells. Comparative RNA-sequencing analysis of different concentrations of MeJA showed that a total of 30, 61, and 39 miRNAs were differentially expressed in the comparisons of CKvsM10, CKvsM50, CKvsM100, respectively. The analysis of the target genes of the differentially expressed miRNAs showed that plant hormone signal transduction, linoleic acid, and alpha-linolenic acid metabolism-related genes were significantly enriched. In addition, we found that miR160a-5p target ARF, miR171d_1 and miR171f_3 target DELLA, miR171b-3p target ETR, and miR156a target BRI1, which played a key role in rosemary suspension cells under MeJA treatments. qRT-PCR of 12 differentially expressed miRNAs and their target genes showed a high correlation between the RNA-seq and the qRT-PCR result. Amplification culture of rosemary suspension cells in a 5 L stirred bioreactor showed that cell biomass accumulation in the bioreactor was less than that in the shake flask under the same conditions, and the whole cultivation period was extended to 14 d. Taken together, MeJA promoted the synthesis of the active compounds in rosemary suspension cells in a wide concentration range via concentration-dependent differential expression patterns. This study provided an overall view of the miRNAs responding to MeJA in rosemary.
Keywords: MeJA; RNA-seq; Rosmarinus officinalis L.; active compounds; miRNAs; stirred bioreactor; suspension cells.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




References
-
- Holmes P. Rosemary oil: The wisdom of the heart. Int. J. Aromather. 1999;9:62–66. doi: 10.1016/S0962-4562(98)80021-0. - DOI
-
- Neto N.J.G., Luz I.D.S., Tavares A.G., Honório V.G., Magnani M., De Souza E.L. Rosmarinus officinalis L. Essential Oil and Its Majority Compound 1,8-Cineole at Sublethal Amounts Induce No Direct and Cross Protection in Staphylococcus aureus ATCC. Foodborne Pathog. Dis. 2012;9:1071–1076. doi: 10.1089/fpd.2012.1258. - DOI - PubMed
-
- Bomfim N.D.S., Nakassugi L.P., Oliveira J.F., Kohiyama C.Y., Mossini S.A., Grespan R., Nerilo S.B., Mallmann C., Abreu Filho B.A., Machinski M., Jr. Antifungal activity and inhibition of fumonisin production by Rosmarinus officinalis L. essential oil in Fusarium verticillioides (Sacc.) Nirenberg. Food Chem. 2015;166:330–336. doi: 10.1016/j.foodchem.2014.06.019. - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

