Necroptosis as a Novel Facet of Mitotic Catastrophe
- PMID: 35409093
- PMCID: PMC8998610
- DOI: 10.3390/ijms23073733
Necroptosis as a Novel Facet of Mitotic Catastrophe
Abstract
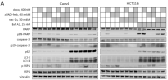
Mitotic catastrophe is a defensive mechanism that promotes elimination of cells with aberrant mitosis by triggering the cell-death pathways and/or cellular senescence. Nowadays, it is known that apoptosis, autophagic cell death, and necrosis could be consequences of mitotic catastrophe. Here, we demonstrate the ability of a DNA-damaging agent, doxorubicin, at 600 nM concentration to stimulate mitotic catastrophe. We observe that the inhibition of caspase activity leads to accumulation of cells with mitotic catastrophe hallmarks in which RIP1-dependent necroptotic cell death is triggered. The suppression of autophagy by a chemical inhibitor or ATG13 knockout upregulates RIP1 phosphorylation and promotes necroptotic cell death. Thus, in certain conditions mitotic catastrophe, in addition to apoptosis and autophagy, can precede necroptosis.
Keywords: autophagy; doxorubicin treatment; mitotic catastrophe; necroptosis.
Conflict of interest statement
The authors declare no conflict of interest.
Figures





Similar articles
-
A link between mitotic defects and mitotic catastrophe: detection and cell fate.Biol Direct. 2021 Dec 9;16(1):25. doi: 10.1186/s13062-021-00313-7. Biol Direct. 2021. PMID: 34886882 Free PMC article. Review.
-
Mitotic catastrophe as a consequence of chemotherapy.Anticancer Agents Med Chem. 2006 Nov;6(6):589-602. doi: 10.2174/187152006778699086. Anticancer Agents Med Chem. 2006. PMID: 17100562 Review.
-
Mitotic catastrophe occurs in the absence of apoptosis in p53-null cells with a defective G1 checkpoint.PLoS One. 2011;6(8):e22946. doi: 10.1371/journal.pone.0022946. Epub 2011 Aug 10. PLoS One. 2011. PMID: 21853057 Free PMC article.
-
Suppression of autophagic flux contributes to cardiomyocyte death by activation of necroptotic pathways.J Mol Cell Cardiol. 2017 Jul;108:203-213. doi: 10.1016/j.yjmcc.2017.06.008. Epub 2017 Jun 21. J Mol Cell Cardiol. 2017. PMID: 28647341
-
Mechanisms of drug-induced mitotic catastrophe in cancer cells.Curr Pharm Des. 2010 Jan;16(1):69-78. doi: 10.2174/138161210789941801. Curr Pharm Des. 2010. PMID: 20214619 Review.
Cited by
-
The 12-Membered TNFR1 Peptide, as Well as the 16-Membered and 6-Membered TNF Peptides, Regulate TNFR1-Dependent Cytotoxic Activity of TNF.Int J Mol Sci. 2024 Mar 31;25(7):3900. doi: 10.3390/ijms25073900. Int J Mol Sci. 2024. PMID: 38612709 Free PMC article.
-
Polyploidy in Cancer: Causal Mechanisms, Cancer-Specific Consequences, and Emerging Treatments.Mol Cancer Ther. 2024 May 2;23(5):638-647. doi: 10.1158/1535-7163.MCT-23-0578. Mol Cancer Ther. 2024. PMID: 38315992 Free PMC article. Review.
-
Antibody-drug conjugates in cancer therapy: mechanisms and clinical studies.MedComm (2020). 2024 Jul 28;5(8):e671. doi: 10.1002/mco2.671. eCollection 2024 Aug. MedComm (2020). 2024. PMID: 39070179 Free PMC article. Review.
-
XMU-MP-1, Inhibitor of STE20-like MST1/2 Kinases of the Hippo Signaling Pathway, Suppresses the Cell Cycle, Activates Apoptosis and Autophagy, and Induces Death of Hematopoietic Tumor Cells.Pharmaceuticals (Basel). 2025 Jun 12;18(6):874. doi: 10.3390/ph18060874. Pharmaceuticals (Basel). 2025. PMID: 40573272 Free PMC article.
-
Special Issue "Advances in Genome Regulation in Cancer".Int J Mol Sci. 2023 Sep 26;24(19):14567. doi: 10.3390/ijms241914567. Int J Mol Sci. 2023. PMID: 37834014 Free PMC article.
References
-
- Salmina K., Bojko A., Inashkina I., Staniak K., Dudkowska M., Podlesniy P., Rumnieks F., Vainshelbaum N.M., Pjanova D., Sikora E., et al. “Mitotic Slippage” and Extranuclear DNA in Cancer Chemoresistance: A Focus on Telomeres. Int. J. Mol. Sci. 2020;21:2779. doi: 10.3390/ijms21082779. - DOI - PMC - PubMed
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

