The Tris(pentafluorophenyl)methylium Cation: Isolation and Reactivity
- PMID: 35416383
- PMCID: PMC9401592
- DOI: 10.1002/anie.202203777
The Tris(pentafluorophenyl)methylium Cation: Isolation and Reactivity
Abstract
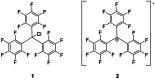
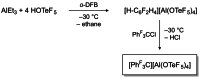
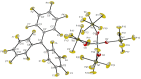
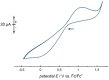
Herein, we present two different routes for the synthesis of the perfluorinated trityl cation, which allowed the handling of the free, uncoordinated species in organic solvents for the first time. The usage of the weakly coordinating anion [Al(OTeF5 )4 ]- and its derivatives allows the characterization of this species by NMR spectroscopy and most importantly by single-crystal X-ray diffraction. The high hydride ion affinity of the cation is shown by hydrogen abstraction from isobutane. Furthermore, cyclic voltammetry reveals its oxidative potential which is supported by the reaction with tris(4-bromophenyl)amine, giving rise to the formation of the ammoniumyl radical cation, also known as "magic blue".
Keywords: High Hydride Affinity; Perfluorinated Trityl Cation; Strong Oxidizer; Superacids; Weakly Coordinating Anions.
© 2022 The Authors. Angewandte Chemie International Edition published by Wiley-VCH GmbH.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



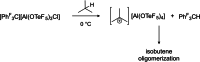

References
-
- None
-
- Chen E. Y., Marks T. J., Chem. Rev. 2000, 100, 1391; - PubMed
-
- McGuinness D. S., Rucklidge A. J., Tooze R. P., Slawin A. M. Z., Organometallics 2007, 26, 2561;
-
- Bah J., Franzén J., Chem. Eur. J. 2014, 20, 1066. - PubMed
-
- Klare H. F. T., Albers L., Süsse L., Keess S., Müller T., Oestreich M., Chem. Rev. 2021, 121, 5889. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

