When killers become thieves: Trogocytosed PD-1 inhibits NK cells in cancer
- PMID: 35417234
- PMCID: PMC9007500
- DOI: 10.1126/sciadv.abj3286
When killers become thieves: Trogocytosed PD-1 inhibits NK cells in cancer
Abstract
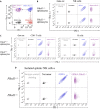
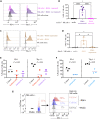
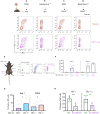
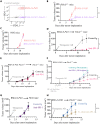
Trogocytosis modulates immune responses, with still unclear underlying molecular mechanisms. Using leukemia mouse models, we found that lymphocytes perform trogocytosis at high rates with tumor cells. While performing trogocytosis, both Natural Killer (NK) and CD8+ T cells acquire the checkpoint receptor PD-1 from leukemia cells. In vitro and in vivo investigation revealed that PD-1 on the surface of NK cells, rather than being endogenously expressed, was derived entirely from leukemia cells in a SLAM receptor-dependent fashion. PD-1 acquired via trogocytosis actively suppressed NK cell antitumor immunity. PD-1 trogocytosis was corroborated in patients with clonal plasma cell disorders, where NK cells that stained for PD-1 also stained for tumor cell markers. Our results, in addition to shedding light on a previously unappreciated mechanism underlying the presence of PD-1 on NK and cytotoxic T cells, reveal the immunoregulatory effect of membrane transfer occurring when immune cells contact tumor cells.
Figures


References
-
- Hudrisier D., Joly E., What is trogocytosis and what is its purpose? Nat. Immunol. 4, 815 (2003). - PubMed
-
- Huang J. F., Yang Y., Sepulveda H., Shi W., Hwang I., Peterson P. A., Jackson M. R., Sprent J., Cai Z., TCR-mediated internalization of peptide-MHC complexes acquired by T cells. Science 286, 952–954 (1999). - PubMed
-
- Hwang I., Huang J. F., Kishimoto H., Brunmark A., Peterson P. A., Jackson M. R., Surh C. D., Cai Z., Sprent J., T cells can use either T cell receptor or CD28 receptors to absorb and internalize cell surface molecules derived from antigen-presenting cells. J. Exp. Med. 191, 1137–1148 (2000). - PMC - PubMed
-
- Hudrisier D., Riond J., Mazarguil H., Gairin J. E., Joly E., Cutting edge: CTLs rapidly capture membrane fragments from target cells in a TCR signaling-dependent manner. J. Immunol. 166, 3645–3649 (2001). - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials

