Weight gain during the dolutegravir transition in the African Cohort Study
- PMID: 35419973
- PMCID: PMC9008168
- DOI: 10.1002/jia2.25899
Weight gain during the dolutegravir transition in the African Cohort Study
Abstract
Introduction: Dolutegravir (DTG) has become a preferred component of first-line antiretroviral therapy (ART) in many settings but may be associated with excess weight gain. We evaluated changes in weight and body mass index (BMI) after switch to single-tablet tenofovir/lamivudine/dolutegravir (TLD) by people living with HIV (PLWH) in four African countries.
Methods: The African Cohort Study (AFRICOS) prospectively follows adults with and without HIV in Kenya, Uganda, Tanzania and Nigeria. Demographics, ART regimen, weight, BMI and waist-to-hip ratio were collected every 6 months. Multivariable Cox proportional hazards modelling was used to estimate hazard ratios and 95% confidence intervals (CIs) for factors associated with developing a BMI ≥25 kg/m2 . Linear mixed effects models with random effects were used to examine the average change in BMI, weight and waist-to-hip ratio.
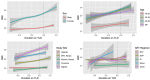
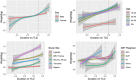
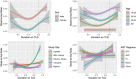
Results: From 23 January 2013 to 1 December 2020, 2950 PLWH were enrolled in AFRICOS and 1474 transitioned to TLD. In adjusted models, PLWH on TLD had 1.77 times the hazard of developing a high BMI (95% CI: 1.22-2.55) compared to PLWH on non-TLD ART. Examining change in weight among all PLWH on ART, participants on TLD gained an average of 0.68 kg (95% CI: 0.32-1.04) more than PLWH on other regimens after adjusting for duration on ART, sex, age, study site and CD4 nadir. Among participants who switched to TLD, the average change in weight prior to TLD switch was 0.35 kg/year (95% CI: 0.25-0.46) and average change in weight was 1.46 kg/year (95% CI: 1.18-1.75) in the year following transition to TLD after adjustment for confounders.
Conclusions: Elevated BMI and weight gain among PLWH on TLD are concerning safety signals. Implications for the development of metabolic comorbidities should be monitored, particularly if annual weight gain persists during continued follow-up after transitioning to TLD.
Keywords: Africa; HIV integrase inhibitors; antiretroviral therapy; body mass index; dolutegravir; weight gain.
© 2022 The Authors. Journal of the International AIDS Society published by John Wiley & Sons Ltd on behalf of the International AIDS Society.
Conflict of interest statement
The authors have no competing of interests to disclose.
Figures
References
-
- Teeraananchai S, Kerr SJ, Amin J, Ruxrungtham K, Law MG. Life expectancy of HIV‐positive people after starting combination antiretroviral therapy: a meta‐analysis. HIV Med. 2017;18(4):256–66. - PubMed
-
- Achhra AC, Sabin C, Ryom L, Hatleberg C, Antonella dʼAminio M, de Wit S, et al. Body mass index and the risk of serious non‐AIDS events and all‐cause mortality in treated HIV‐positive individuals: D: A: D cohort analysis. J Acquir Immune Defic Syndr. 2018;78(5):579–88. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

