Fungal CNS Infections in Africa: The Neuroimmunology of Cryptococcal Meningitis
- PMID: 35432326
- PMCID: PMC9010970
- DOI: 10.3389/fimmu.2022.804674
Fungal CNS Infections in Africa: The Neuroimmunology of Cryptococcal Meningitis
Abstract
Cryptococcal meningitis (CM) is the leading cause of central nervous system (CNS) fungal infections in humans, with the majority of cases reported from the African continent. This is partly due to the high burden of HIV infection in the region and reduced access to standard-of-care including optimal sterilising antifungal drug treatments. As such, CM is responsible for 10-15% of all HIV-related mortality, with a large proportion being preventable. Immunity to the causative agent of CM, Cryptococcus neoformans, is only partially understood. IFNγ producing CD4+ T-cells are required for the activation of myeloid cells, especially macrophages, to enable fungal killing and clearance. However, macrophages may also act as a reservoir of the fungal yeast cells, shielding them from host immune detection thus promoting latent infection or persistent chronic inflammation. In this chapter, we review the epidemiology and pathogenesis of CNS fungal infections in Africa, with a major focus on CM, and the antifungal immune pathways operating to protect against C. neoformans infection. We also highlight the areas of research and policy that require prioritisation to help reduce the burden of CNS fungal diseases in Africa.
Keywords: HAART; astrocyte; cryptococcal meningitis; fungal infection; microglia.
Copyright © 2022 Mohamed, Nyazika, Ssebambulidde, Lionakis, Meya and Drummond.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
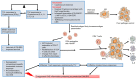

References
-
- Spina-Tensini T, Dominguez Muro M, Queiroz-Telles F, Strozzi I, Talise Moraes S, Petterle RR, et al. . Geographic Distribution of Patients Affected by Cryptococcus Neoformans/Cryptococcus Gattii Species Complexes Meningitis, Pigeon and Tree Populations in Southern Brazil. Mycoses (2017) 60(1):51–8. doi: 10.1111/myc.12550 - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

