The ArcAB Two-Component System: Function in Metabolism, Redox Control, and Infection
- PMID: 35442087
- PMCID: PMC9199408
- DOI: 10.1128/mmbr.00110-21
The ArcAB Two-Component System: Function in Metabolism, Redox Control, and Infection
Abstract
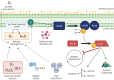
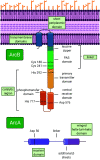
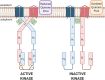
ArcAB, also known as the Arc system, is a member of the two-component system family of bacterial transcriptional regulators and is composed of sensor kinase ArcB and response regulator ArcA. In this review, we describe the structure and function of these proteins and assess the state of the literature regarding ArcAB as a sensor of oxygen consumption. The bacterial quinone pool is the primary modulator of ArcAB activity, but questions remain for how this regulation occurs. This review highlights the role of quinones and their oxidation state in activating and deactivating ArcB and compares competing models of the regulatory mechanism. The cellular processes linked to ArcAB regulation of central metabolic pathways and potential interactions of the Arc system with other regulatory systems are also reviewed. Recent evidence for the function of ArcAB under aerobic conditions is challenging the long-standing characterization of this system as strictly an anaerobic global regulator, and the support for additional ArcAB functionality in this context is explored. Lastly, ArcAB-controlled cellular processes with relevance to infection are assessed.
Keywords: facultative anaerobes; global regulatory networks; metabolic regulation; metabolism; two-component regulatory systems.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
-
- Ward B. 2015. Bacterial energy metabolism, p 201–233. In Tang Y-W, Sussman M, Liu D, Poxton I, Schwartzman J (ed), Molecular medical microbiology, 2nd ed. Academic Press, Boston, MA.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

