Early post-infection treatment of SARS-CoV-2 infected macaques with human convalescent plasma with high neutralizing activity had no antiviral effects but moderately reduced lung inflammation
- PMID: 35443018
- PMCID: PMC9060337
- DOI: 10.1371/journal.ppat.1009925
Early post-infection treatment of SARS-CoV-2 infected macaques with human convalescent plasma with high neutralizing activity had no antiviral effects but moderately reduced lung inflammation
Abstract
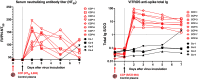
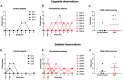
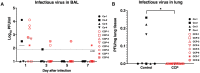
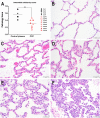
Early in the SARS-CoV-2 pandemic, there was a high level of optimism based on observational studies and small controlled trials that treating hospitalized patients with convalescent plasma from COVID-19 survivors (CCP) would be an important immunotherapy. However, as more data from controlled trials became available, the results became disappointing, with at best moderate evidence of efficacy when CCP with high titers of neutralizing antibodies was used early in infection. To better understand the potential therapeutic efficacy of CCP, and to further validate SARS-CoV-2 infection of macaques as a reliable animal model for testing such strategies, we inoculated 12 adult rhesus macaques with SARS-CoV-2 by intratracheal and intranasal routes. One day later, 8 animals were infused with pooled human CCP with a high titer of neutralizing antibodies (RVPN NT50 value of 3,003), while 4 control animals received normal human plasma. Animals were monitored for 7 days. Animals treated with CCP had detectable but low levels of antiviral antibodies after infusion. In comparison to the control animals, CCP-treated animals had similar levels of viral RNA in upper and lower respiratory tract secretions, similar detection of viral RNA in lung tissues by in situ hybridization, but lower amounts of infectious virus in the lungs. CCP-treated animals had a moderate, but statistically significant reduction in interstitial pneumonia, as measured by comprehensive lung histology. Thus overall, therapeutic benefits of CCP were marginal and inferior to results obtained earlier with monoclonal antibodies in this animal model. By highlighting strengths and weaknesses, data of this study can help to further optimize nonhuman primate models to provide proof-of-concept of intervention strategies, and guide the future use of convalescent plasma against SARS-CoV-2 and potentially other newly emerging respiratory viruses.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures



Update of
-
Early post-infection treatment of SARS-CoV-2 infected macaques with human convalescent plasma with high neutralizing activity reduces lung inflammation.bioRxiv [Preprint]. 2021 Sep 1:2021.09.01.458520. doi: 10.1101/2021.09.01.458520. bioRxiv. 2021. Update in: PLoS Pathog. 2022 Apr 20;18(4):e1009925. doi: 10.1371/journal.ppat.1009925. PMID: 34494025 Free PMC article. Updated. Preprint.
References
-
- Johns Hopkins University of Medicine. Coronavirus Resource Center [cited 2021 February 23]. https://coronavirus.jhu.edu/.
-
- Mair-Jenkins J, Saavedra-Campos M, Baillie JK, Cleary P, Khaw FM, Lim WS, et al. The effectiveness of convalescent plasma and hyperimmune immunoglobulin for the treatment of severe acute respiratory infections of viral etiology: a systematic review and exploratory meta-analysis. J Infect Dis. 2015;211(1):80–90. Epub 2014/07/18. doi: 10.1093/infdis/jiu396 . - DOI - PMC - PubMed
-
- FDA issues Emergency Use Authorization for Convalescent Plasma as potential promising COVID–19 Treatment, Another achievement in administration’s fight against pandemic [Internet]. August 23, 2020. https://www.fda.gov/news-events/press-announcements/fda-issues-emergency...
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

