Impaired phosphate transport in SLC34A2 variants in patients with pulmonary alveolar microlithiasis
- PMID: 35443721
- PMCID: PMC9019944
- DOI: 10.1186/s40246-022-00387-y
Impaired phosphate transport in SLC34A2 variants in patients with pulmonary alveolar microlithiasis
Abstract
Background: Variants in SLC34A2 encoding the sodium-dependent phosphate transport protein 2b (NaPi-IIb) cause the rare lung disease pulmonary alveolar microlithiasis (PAM). PAM is characterised by the deposition of calcium-phosphate concretions in the alveoli usually progressing over time. No effective treatment is available. So far, 30 allelic variants in patients have been reported but only a few have been functionally characterised. This study aimed to determine the impact of selected SLC34A2 variants on transporter expression and phosphate uptake in cellular studies.
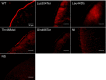
Methods: Two nonsense variants (c.910A > T and c.1456C > T), one frameshift (c.1328delT), and one in-frame deletion (c.1402_1404delACC) previously reported in patients with PAM were selected for investigation. Wild-type and mutant c-Myc-tagged human NaPi-IIb constructs were expressed in Xenopus laevis oocytes. The transport function was investigated with a 32Pi uptake assay. NaPi-IIb protein expression and localisation were determined with immunoblotting and immunohistochemistry, respectively.
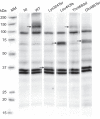
Results: Oocytes injected with the wild-type human NaPi-IIb construct had significant 32Pi transport compared to water-injected oocytes. In addition, the protein had a molecular weight as expected for the glycosylated form, and it was readily detectable in the oocyte membrane. Although the protein from the Thr468del construct was synthesised and expressed in the oocyte membrane, phosphate transport was similar to non-injected control oocytes. All other mutants were non-functional and not expressed in the membrane, consistent with the expected impact of the truncations caused by premature stop codons.
Conclusions: Of four analysed SLC34A2 variants, only the Thr468del showed similar protein expression as the wild-type cotransporter in the oocyte membrane. All mutant transporters were non-functional, supporting that dysfunction of NaPi-IIb underlies the pathology of PAM.
Keywords: In-frame deletion, frameshift variant, nonsense variant; Pulmonary alveolar microlithiasis (PAM); SLC34A2; SLC34A2 mutations; SLC34A2 variants; Sodium-dependent phosphate transport protein 2B, NaPi-2b; Sodium-dependent phosphate transport protein 2B, NaPi-IIb; Xenopus laevis oocytes.
© 2022. The Author(s).
Conflict of interest statement
CAW received honoraria from Medice, Germany, and from Bayer Pharmaceuticals AG, Germany as well as a research grant from Chugai, Japan. The other authors declare that they have no competing interests.
Figures



Similar articles
-
New insights in the genetic variant spectrum of SLC34A2 in pulmonary alveolar microlithiasis; a systematic review.Orphanet J Rare Dis. 2023 May 31;18(1):130. doi: 10.1186/s13023-023-02712-7. Orphanet J Rare Dis. 2023. PMID: 37259144 Free PMC article.
-
Eight novel variants in the SLC34A2 gene in pulmonary alveolar microlithiasis.Eur Respir J. 2020 Feb 27;55(2):1900806. doi: 10.1183/13993003.00806-2019. Print 2020 Feb. Eur Respir J. 2020. PMID: 31831582
-
SLC34A2 Gene mutation of pulmonary alveolar microlithiasis: report of four cases and review of literatures.Respir Med. 2013 Feb;107(2):217-22. doi: 10.1016/j.rmed.2012.10.016. Epub 2012 Nov 17. Respir Med. 2013. PMID: 23164546 Review.
-
A pedigree with pulmonary alveolar microlithiasis: a clinical case report and literature review.Cell Biochem Biophys. 2014 Sep;70(1):565-72. doi: 10.1007/s12013-014-9957-9. Cell Biochem Biophys. 2014. PMID: 24817643
-
Novel deletion of SLC34A2 in Chinese patients of PAM shares mutation hot spot with fusion gene SLC34A2-ROS1 in lung cancer.J Genet. 2018 Sep;97(4):939-944. J Genet. 2018. PMID: 30262706
Cited by
-
New insights in the genetic variant spectrum of SLC34A2 in pulmonary alveolar microlithiasis; a systematic review.Orphanet J Rare Dis. 2023 May 31;18(1):130. doi: 10.1186/s13023-023-02712-7. Orphanet J Rare Dis. 2023. PMID: 37259144 Free PMC article.
-
A Case Report of Pulmonary Alveolar Microlithiasis: Focus on Radiologic Findings.Am J Case Rep. 2023 Jan 7;24:e938456. doi: 10.12659/AJCR.938456. Am J Case Rep. 2023. PMID: 36609435 Free PMC article.
-
Soft tissue calcifications in chronic kidney disease-beyond the vasculature.Pflugers Arch. 2025 Aug;477(8):1037-1059. doi: 10.1007/s00424-025-03098-0. Epub 2025 Jun 5. Pflugers Arch. 2025. PMID: 40471241 Free PMC article. Review.
References
-
- SLC34A2, Gene ID: 10568 [Internet]. Bethesda (MD): National Library of Medicine (US), National Center for Biotechnology Information; 2004—[cited 2021 12 29]. Available from: https://www.ncbi.nlm.nih.gov/gene/.
Publication types
MeSH terms
Substances
Supplementary concepts
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

