miR-34a-5p inhibits the malignant progression of KSHV-infected SH-SY5Y cells by targeting c-fos
- PMID: 35444864
- PMCID: PMC9014853
- DOI: 10.7717/peerj.13233
miR-34a-5p inhibits the malignant progression of KSHV-infected SH-SY5Y cells by targeting c-fos
Abstract
Background: We aimed to investigate the effects of miR-34a-5p on c-fos regulation mediating the malignant behaviors of SH-SY5Y cells infected with Kaposi's sarcoma-associated herpesvirus (KSHV).
Methods: The KSHV-infected (SK-RG) and uninfected SH-SY5Y parent cells were compared for differentially expressed miRNAs using transcriptome sequencing. Then miR-34a-5p was upregulated in SK-RG cells by the miRNA mimics transfection. Cell proliferation ability was determined by MTT and plate clone assays. The cell cycle was assessed by flow cytometry analysis, and CDK4, CDK6, cyclin D1 levels were determined by Western blot analysis. The migration behavior was detected by wound healing and transwell assays. The protein levels of MMP2 and MMP9 were measured by Western blot analysis. The regulation of c-fos by miR-34a-5p was detected by the dual-luciferase reporter gene assay. Rescue assays were carried out by upregulating c-fos in miR-34a-5p-overexpressing SK-RG cells. KSHV DNA copy numbers and relative virus gene expressions were detected. Xenograft tumor experiments and immunohistochemistry assays were further used to detect the effects of miR-34a-5p.
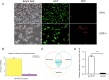
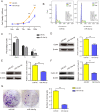
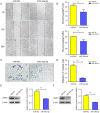
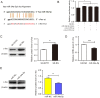
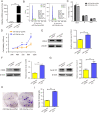
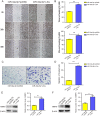
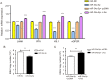
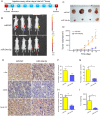
Results: miR-34a-5p was lower in SK-RG cells. Restoration of miR-34a-5p decreased cell proliferation and migration, leading to a G1 cell cycle arrest and down-regulation of CDK4/6, cyclin D1, MMP2, MMP9. KSHV copy number and expression of virus gene including latency-associated nuclear antigen (LANA), replication and transcription activator (RTA), open reading frame (K8.1), and KSHV G protein-coupled receptor (v-GPCR) were also reduced. Furthermore, c-fos is the target of miR-34a-5p, while enhanced c-fos weakened cellular behaviors of miR-34a-5p-overexpressing cells. Xenograft experiments and immunohistochemistry assays showed that miR-34a-5p inhibited tumor growth and virus gene expression.
Conclusion: Upregulated miR-34a-5p in KSHV-infected SH-SY5Y cells suppressed cell proliferation and migration through down-regulating c-fos. miR-34a-5p was a candidate molecular drug for KSHV-infected neuronal cells.
Keywords: Kaposi’s sarcoma–associated herpesvirus; Migration; Proliferation; c-fos; miR-34a-5p.
© 2022 Wu et al.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

