Unusual suspects: Glial cells in fertility regulation and their suspected role in polycystic ovary syndrome
- PMID: 35445462
- PMCID: PMC9489003
- DOI: 10.1111/jne.13136
Unusual suspects: Glial cells in fertility regulation and their suspected role in polycystic ovary syndrome
Abstract
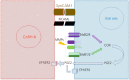
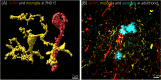
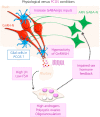
Gonadotropin-releasing-hormone (GnRH) neurons sitting within the hypothalamus control the production of gametes and sex steroids by the gonads, therefore ensuring survival of species. As orchestrators of reproductive function, GnRH neurons integrate information from external and internal cues. This occurs through an extensively studied neuronal network known as the "GnRH neuronal network." However, the brain is not simply composed of neurons. Evidence suggests a role for glial cells in controlling GnRH neuron activity, secretion and fertility outcomes, although numerous questions remain. Glial cells have historically been seen as support cells for neurons. This idea has been challenged by the discovery that some neurological diseases originate from glial dysfunction. The prevalence of infertility disorders is increasing worldwide, with one in four couples being affected; therefore, it remains essential to understand the mechanisms by which the brain controls fertility. The "GnRH glial network" could be a major player in infertility disorders and represent a potential therapeutic target. In polycystic ovary syndrome (PCOS), the most common infertility disorder of reproductive aged women worldwide, the brain is considered a prime suspect. Recent studies have demonstrated pathological neuronal wiring of the "GnRH neuronal network" in PCOS-like animal models. However, the role of the "GnRH glial network" remains to be elucidated. In this review, I aim to propose glial cells as unusual suspects in infertility disorders such as PCOS. In the first part, I state our current knowledge about the role of glia in the regulation of GnRH neurons and fertility. In the second part, based on our recent findings, I discuss how glial cells could be implicated in PCOS pathology.
Keywords: GnRH; PCOS; astrocyte; fertility; glia; microglia; tanycyte.
© 2022 British Society for Neuroendocrinology.
Conflict of interest statement
The author declares that she has no conflicts of interests.
Figures


Similar articles
-
The role of gonadotropin-releasing hormone neurons in polycystic ovary syndrome.J Neuroendocrinol. 2022 May;34(5):e13093. doi: 10.1111/jne.13093. Epub 2022 Jan 26. J Neuroendocrinol. 2022. PMID: 35083794 Free PMC article. Review.
-
Impaired steroid hormone feedback in polycystic ovary syndrome: Evidence from preclinical models for abnormalities within central circuits controlling fertility.Clin Endocrinol (Oxf). 2022 Aug;97(2):199-207. doi: 10.1111/cen.14711. Epub 2022 Mar 29. Clin Endocrinol (Oxf). 2022. PMID: 35349177 Free PMC article. Review.
-
Prepubertal Development of GABAergic Transmission to Gonadotropin-Releasing Hormone (GnRH) Neurons and Postsynaptic Response Are Altered by Prenatal Androgenization.J Neurosci. 2018 Feb 28;38(9):2283-2293. doi: 10.1523/JNEUROSCI.2304-17.2018. Epub 2018 Jan 26. J Neurosci. 2018. PMID: 29374136 Free PMC article.
-
Activation of arcuate nucleus GABA neurons promotes luteinizing hormone secretion and reproductive dysfunction: Implications for polycystic ovary syndrome.EBioMedicine. 2019 Jun;44:582-596. doi: 10.1016/j.ebiom.2019.05.065. Epub 2019 Jun 6. EBioMedicine. 2019. PMID: 31178425 Free PMC article.
-
Morphological evidence indicates a role for microglia in shaping the PCOS-like brain.J Neuroendocrinol. 2021 Aug;33(8):e12999. doi: 10.1111/jne.12999. Epub 2021 Jul 3. J Neuroendocrinol. 2021. PMID: 34216402
Cited by
-
SEMA6A drives GnRH neuron-dependent puberty onset by tuning median eminence vascular permeability.Nat Commun. 2023 Dec 7;14(1):8097. doi: 10.1038/s41467-023-43820-z. Nat Commun. 2023. PMID: 38062045 Free PMC article.
-
Investigation of the Effects of Metallic Nanoparticles on Fertility Outcomes and Endocrine Modification of the Hypothalamic-Pituitary-Gonadal Axis.Int J Mol Sci. 2023 Jul 20;24(14):11687. doi: 10.3390/ijms241411687. Int J Mol Sci. 2023. PMID: 37511445 Free PMC article.
-
New perspectives on polycystic ovary syndrome: hypothalamic-sympathetic-adipose tissue interaction.J Ovarian Res. 2025 Jul 4;18(1):145. doi: 10.1186/s13048-025-01724-0. J Ovarian Res. 2025. PMID: 40615863 Free PMC article. Review.
References
-
- Herbison AE. Control of puberty onset and fertility by gonadotropin‐releasing hormone neurons. Nat Rev Endocrinol. 2016;12(8):452‐466. - PubMed
-
- Herbison AE. A simple model of estrous cycle negative and positive feedback regulation of GnRH secretion. Front Neuroendocrinol. 2020;57:100837. - PubMed
-
- Azevedo FA, Carvalho LR, Grinberg LT, et al. Equal numbers of neuronal and nonneuronal cells make the human brain an isometrically scaled‐up primate brain. J Comp Neurol. 2009;513(5):532‐541. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

