A Combined Conduit-Bioactive Hydrogel Approach for Regeneration of Transected Sciatic Nerves
- PMID: 35446025
- PMCID: PMC11097895
- DOI: 10.1021/acsabm.2c00132
A Combined Conduit-Bioactive Hydrogel Approach for Regeneration of Transected Sciatic Nerves
Abstract
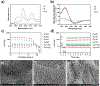
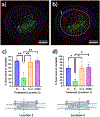
Transected peripheral nerve injury (PNI) affects the quality of life of patients, which leads to socioeconomic burden. Despite the existence of autografts and commercially available nerve guidance conduits (NGCs), the complexity of peripheral nerve regeneration requires further research in bioengineered NGCs to improve surgical outcomes. In this work, we introduce multidomain peptide (MDP) hydrogels, as intraluminal fillers, into electrospun poly(ε-caprolactone) (PCL) conduits to bridge 10 mm rat sciatic nerve defects. The efficacy of treatment groups was evaluated by electromyography and gait analysis to determine their electrical and motor recovery. We then studied the samples' histomorphometry with immunofluorescence staining and automatic axon counting/measurement software. Comparison with negative control group shows that PCL conduits filled with an anionic MDP may improve functional recovery 16 weeks postoperation, displaying higher amplitude of compound muscle action potential, greater gastrocnemius muscle weight retention, and earlier occurrence of flexion contracture. In contrast, PCL conduits filled with a cationic MDP showed the least degree of myelination and poor functional recovery. This phenomenon may be attributed to MDPs' difference in degradation time. Electrospun PCL conduits filled with an anionic MDP may become an attractive tissue engineering strategy for treating transected PNI when supplemented with other bioactive modifications.
Keywords: electromyography; nerve guidance conduit; peptide; peripheral nerve regeneration; sciatic nerve transection; self-assembly.
Conflict of interest statement
The authors declare no competing financial interest.
Figures









References
-
- Eser F; Aktekin L; Bodur H; Atan Ç Etiological Factors of Traumatic Peripheral Nerve Injuries. Neurol. India 2009, 57 (4), 434–437. - PubMed
-
- Taylor CA; Braza D; Rice JB; Dillingham T The Incidence of Peripheral Nerve Injury in Extremity Trauma. Am. J. Phys. Med. Rehabil 2008, 87 (5), 381–385. - PubMed
-
- Raza C; Riaz HA; Anjum R; Shakeel NUA Repair Strategies for Injured Peripheral Nerve: Review. Life Sci. 2020, 243, 117308. - PubMed
-
- Kehoe S; Zhang XF; Boyd D FDA Approved Guidance Conduits and Wraps for Peripheral Nerve Injury: A Review of Materials and Efficacy. Injury 2012, 43 (5), 553–572. - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
