Real-world impact of primary immune thrombocytopenia and treatment with thrombopoietin receptor agonists on quality of life based on patient-reported experience: Results from a questionnaire conducted in Switzerland, Austria, and Belgium
- PMID: 35446925
- PMCID: PMC9022837
- DOI: 10.1371/journal.pone.0267342
Real-world impact of primary immune thrombocytopenia and treatment with thrombopoietin receptor agonists on quality of life based on patient-reported experience: Results from a questionnaire conducted in Switzerland, Austria, and Belgium
Abstract
Aims of the study: Thrombopoietin receptor agonists (TPO-RAs) are approved for immune thrombocytopenia (ITP), but their impact on health-related quality of life (HRQoL) remains poorly investigated in clinical practice. This observational study aimed to gain insight into real-world patient-reported experiences of the burden of ITP and TPO-RAs.
Method: An online questionnaire of closed questions was used to collect views of patients with primary ITP from Switzerland, Austria, and Belgium, between September 2018 and April 2020.
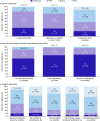
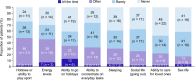
Results: Of 46 patients who completed the questionnaire (total cohort), 41% were receiving TPO-RAs. A numerically higher proportion of patients reported being free from symptoms at the time of the questionnaire (54%) than at diagnosis (24%), irrespective of treatment type. Bleeding, the most frequently reported symptom at diagnosis (59%), was reduced at the time of the questionnaire (7%). Conversely, fatigue was reported by approximately 40% of patients at both diagnosis and the time of the questionnaire. Having a normal life and their disease under control was reported by 83% and 76%, respectively, but 41% were worried/anxious about their condition. Nearly 50% reported that ITP impaired their engagement in hobbies/sport or energy levels and 63% reported no impact on employment. When stratified by TPO-RA use, bleeding was better controlled in those receiving TPO-RAs than not (0% vs 11%). A numerically lower proportion receiving TPO-RAs than not reported worry/anxiety about their condition (16% vs 59%) and shifting from full-time to part-time employment (11% vs 22%). Similar proportions were satisfied with their therapy whether they were receiving TPO-RAs or not (89% vs 85%).
Conclusions: Many factors affect HRQoL in patients with ITP. Of patients receiving TPO-RAs, none experienced bleeding at the time of the questionnaire; they also showed a more positive perspective for some outcomes than those not using TPO-RAs. However, fatigue was not reduced by any treatment.
Conflict of interest statement
ARovó was not paid for her work on this study. She has acted as a consultant, taken part in advisory boards, and received honoraria and research funding from Novartis. She has also acted as a consultant, taken part in advisory boards, and received honoraria and/or research funding from Alexion, AstraZeneca, Celgene, CSL Behring, and OrPha Swiss. NC was not paid for his work on the study. He has acted as a consultant for Celgene and Novartis and has received honoraria (consultancy honoraria and travel grants) from AbbVie, Alexion, Amgen, BMS, Celgene, Gilead, Incyte, Janssen-Cilag, Novartis, OrPha Swiss, Pfizer, Roche, Sandoz, Sanofi-Aventis, Shire, Takeda, as well as research funding from AstraZeneca, CSL Behring, Novartis, Pierre Fabre, and Shire (Baxter Oncology). KS was not paid for his work on this study. He has acted as a consultant and taken part in advisory boards organized by Novartis. He has also taken part in advisory boards organized by Alexion, Celgene, Janssen-Cilag, OrPha Swiss, and Takeda. ARüfer was not paid for his work on the study. He has taken part in advisory boards organized by Amgen, BMS/Celgene, Janssen-Cilag, Novartis, OrPha Swiss, and Takeda. GK was an employee of Novartis Pharma Schweiz AG at the time of the study, and is now an employee of Novartis AG. SI, and DC are employees of Novartis Pharma Schweiz AG. RB was not paid for his work on the study. He has acted as a consultant, taken part in advisory boards, and received research funding from Novartis. He has also taken part in advisory boards and received honoraria and travel support to educational events from AbbVie, Alexion, Celgene, OrPha Swiss, and Takeda. This does not alter our adherence to PLOS ONE policies on sharing data and materials.
Figures


Similar articles
-
Sustained remission of chronic immune thrombocytopenia after discontinuation of treatment with thrombopoietin-receptor agonists in adults.Int J Hematol. 2015 Jul;102(1):7-11. doi: 10.1007/s12185-015-1793-1. Epub 2015 Apr 2. Int J Hematol. 2015. PMID: 25833724
-
Thrombopoietin Receptor Agonists in Children with Immune Thrombocytopenia: A New Therapeutic Era.Endocr Metab Immune Disord Drug Targets. 2021;21(3):397-406. doi: 10.2174/1871530320666200531142244. Endocr Metab Immune Disord Drug Targets. 2021. PMID: 32473624 Review.
-
The use of thrombopoietin-receptor agonists (TPO-RAs) in immune thrombocytopenia (ITP): a "real life" retrospective multicenter experience of the Rete Ematologica Pugliese (REP).Ann Hematol. 2016 Jan;95(2):239-44. doi: 10.1007/s00277-015-2556-z. Epub 2015 Nov 24. Ann Hematol. 2016. PMID: 26596973
-
Treatment-free remission after thrombopoietin receptor agonist discontinuation in patients with newly diagnosed immune thrombocytopenia: an observational retrospective analysis in real-world clinical practice.Int J Hematol. 2020 Aug;112(2):159-168. doi: 10.1007/s12185-020-02893-y. Epub 2020 May 31. Int J Hematol. 2020. PMID: 32476083
-
Use of thrombopoietin receptor agonists in adults with immune thrombocytopenia: a systematic review and Central European expert consensus.Ann Hematol. 2023 Apr;102(4):715-727. doi: 10.1007/s00277-023-05114-8. Epub 2023 Feb 24. Ann Hematol. 2023. PMID: 36826482 Free PMC article.
Cited by
-
Health-related quality of life and complications of corticosteroid treatment in patients with immune thrombocytopenia in two teaching hospitals in Ethiopia: a cross-sectional study.Front Med (Lausanne). 2024 Nov 5;11:1423161. doi: 10.3389/fmed.2024.1423161. eCollection 2024. Front Med (Lausanne). 2024. PMID: 39564500 Free PMC article.
-
A Qualitative Study on Patient Experience with Signs, Symptoms, and Daily Impacts of Immune Thrombocytopenia.Patient. 2025 Aug 16. doi: 10.1007/s40271-025-00762-6. Online ahead of print. Patient. 2025. PMID: 40817963
-
Spotlight on eltrombopag concentration in pediatric immune thrombocytopenia: A single-center observational study in China.Pediatr Investig. 2024 Jan 29;8(1):44-52. doi: 10.1002/ped4.12411. eCollection 2024 Mar. Pediatr Investig. 2024. PMID: 38516133 Free PMC article.
-
Burden of immune thrombocytopenia (ITP): Special considerations for refractory ITP.Br J Haematol. 2023 Oct;203(1):79-85. doi: 10.1111/bjh.19068. Br J Haematol. 2023. PMID: 37735553 Free PMC article. Review.
-
Impact of Primary Chronic Immune Thrombocytopenia and Thrombopoietin Receptor Agonists Treatment Instructions on Daily Living: Results of a Multinational Cross-Sectional Survey.Patient Prefer Adherence. 2025 Aug 13;19:2445-2458. doi: 10.2147/PPA.S505337. eCollection 2025. Patient Prefer Adherence. 2025. PMID: 40827208 Free PMC article.
References
-
- Rodeghiero F, Stasi R, Gernsheimer T, Michel M, Provan D, Arnold DM, et al.. Standardization of terminology, definitions and outcome criteria in immune thrombocytopenic purpura of adults and children: report from an international working group. Blood. 2009;113(11):2386–93. Epub 2008/11/14. doi: 10.1182/blood-2008-07-162503 . - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

