Phytochemical Profiling and Isolation of Bioactive Compounds from Leucosidea sericea (Rosaceae)
- PMID: 35449904
- PMCID: PMC9016878
- DOI: 10.1021/acsomega.2c00096
Phytochemical Profiling and Isolation of Bioactive Compounds from Leucosidea sericea (Rosaceae)
Abstract
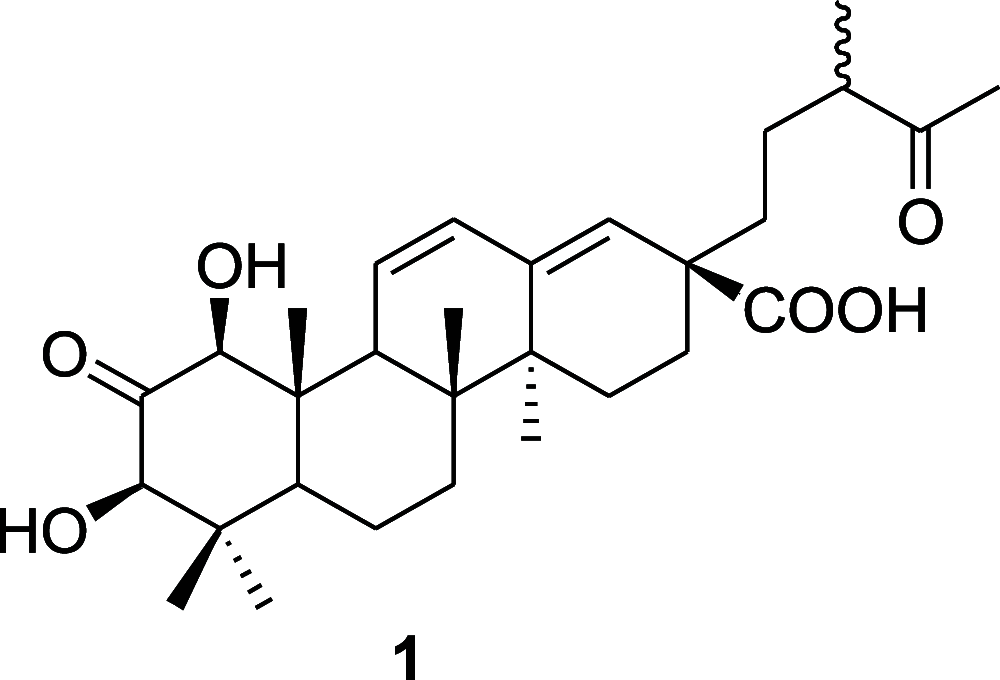
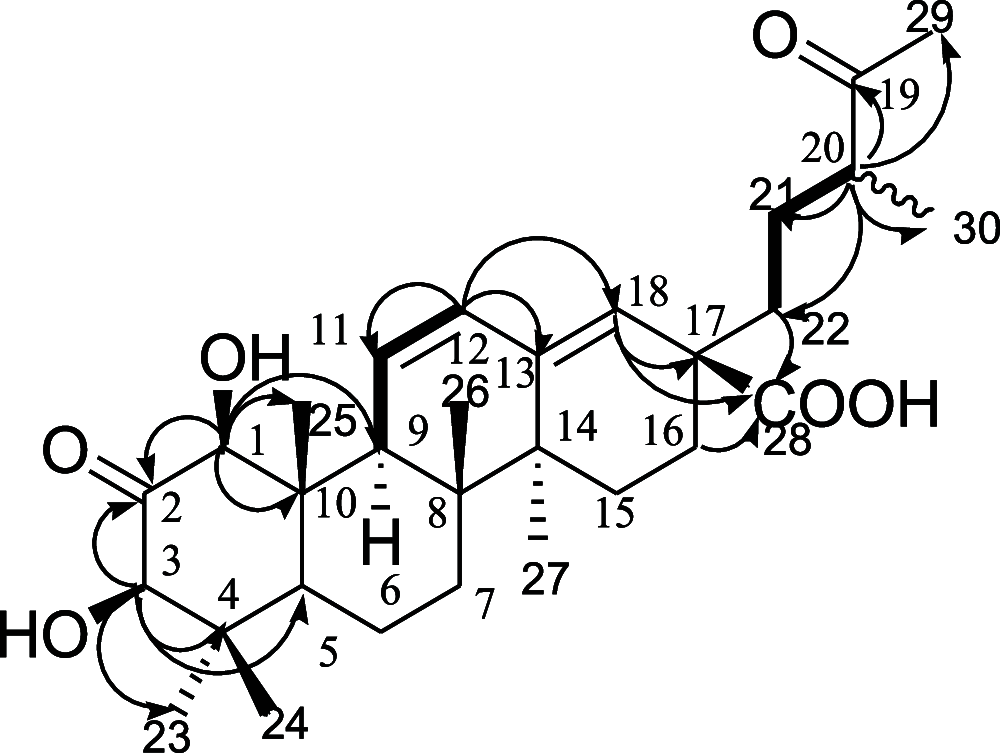
In the study, ultraperformance liquid chromatography-quadrupole time-of-flight-mass spectrometry analysis of Leucosidea sericea leaf and stem extracts led to the identification of various classes of compounds. Further chromatographic purifications resulted in the isolation of 22 compounds that consisted of a new triterpenoid named leucosidic acid A (1), an acetophenone derivative 2, a phloroglucinol derivative 3, three chromones 4-6, seven pentacyclic triterpenoids 7-13, a phytosterol glucoside 14, a flavonoid 15, and seven flavonoid glycosides 16-22. Nineteen of these compounds including the previously undescribed triterpenoid 1 are isolated for the first time from L. sericea. The structures of the isolated compounds were assigned based on their high-resolution mass spectrometry and nuclear magnetic resonance data. Some of the isolated triterpenoids were evaluated for inhibitory activity against α-amylase, α-glucosidase, and pancreatic lipase. Of the tested compounds, 1-hydroxy-2-oxopomolic acid (7) and pomolic acid (13) showed higher potency on α-glucosidase than acarbose, which is used as a positive control in this study. The two compounds inhibited α-glucosidase with IC50 values of 192.1 ± 13.81 and 85.5 ± 6.87 μM, respectively.
© 2022 The Authors. Published by American Chemical Society.
Conflict of interest statement
The authors declare no competing financial interest.
Figures
Similar articles
-
Identification and characterization of potential bioactive compounds from the leaves of Leucosidea sericea.J Ethnopharmacol. 2018 Jun 28;220:169-176. doi: 10.1016/j.jep.2018.03.035. Epub 2018 Mar 28. J Ethnopharmacol. 2018. PMID: 29604376
-
An overview on Leucosidea sericea Eckl. & Zeyh.: A multi-purpose tree with potential as a phytomedicine.J Ethnopharmacol. 2017 May 5;203:288-303. doi: 10.1016/j.jep.2017.03.044. Epub 2017 Mar 28. J Ethnopharmacol. 2017. PMID: 28363524 Review.
-
Ultrastructure changes induced by the phloroglucinol derivative agrimol G isolated from Leucosidea sericea in Haemonchus contortus.Exp Parasitol. 2019 Dec;207:107780. doi: 10.1016/j.exppara.2019.107780. Epub 2019 Oct 17. Exp Parasitol. 2019. PMID: 31629699
-
Quadruple high-resolution α-glucosidase/α-amylase/PTP1B/radical scavenging profiling combined with high-performance liquid chromatography-high-resolution mass spectrometry-solid-phase extraction-nuclear magnetic resonance spectroscopy for identification of antidiabetic constituents in crude root bark of Morus alba L.J Chromatogr A. 2018 Jun 29;1556:55-63. doi: 10.1016/j.chroma.2018.04.041. Epub 2018 Apr 21. J Chromatogr A. 2018. PMID: 29729863
-
Chemical Constituents of Malaysian U. cordata var. ferruginea and Their in Vitro α-Glucosidase Inhibitory Activities.Molecules. 2016 Apr 27;21(5):525. doi: 10.3390/molecules21050525. Molecules. 2016. PMID: 27128898 Free PMC article.
Cited by
-
In vitro biological activities of silver nanoparticles using methanolic leaf extract of Plumeria rubra.Sci Rep. 2025 Aug 3;15(1):28303. doi: 10.1038/s41598-025-14353-w. Sci Rep. 2025. PMID: 40754561 Free PMC article.
-
Bioactive Compounds from Medicinal Plants as Potential Adjuvants in the Treatment of Mild Acne Vulgaris.Molecules. 2024 May 19;29(10):2394. doi: 10.3390/molecules29102394. Molecules. 2024. PMID: 38792254 Free PMC article. Review.
-
Two Triterpenoids, ARM-2 and RA-5, From Protorhus longifolia Exhibit the Potential to Modulate Lipolysis and Lipogenesis in Cultured 3T3-L1 Adipocytes.J Lipids. 2024 Oct 17;2024:3972941. doi: 10.1155/2024/3972941. eCollection 2024. J Lipids. 2024. PMID: 39450349 Free PMC article.
-
Arazá: Eugenia stipitata Mc Vaught as a Potential Functional Food.Foods. 2024 Jul 23;13(15):2310. doi: 10.3390/foods13152310. Foods. 2024. PMID: 39123500 Free PMC article. Review.
References
-
- Shulaev V.; Korban S. S.; Sosinski B.; Abbott A. G.; Aldwinckle H. S.; Folta K. M.; Iezzoni A.; Main D.; Arús P.; Dandekar A. M.; Lewers K.; Brown S. K.; Davis T. M.; Gardiner S. E.; Potter D.; Veilleux R. E. Multiple Models for Rosaceae Genomics. Plant Physiol. 2008, 147, 985–1003. 10.1104/pp.107.115618. - DOI - PMC - PubMed
-
- Hummer K. E.; Janick J.. Rosaceae: Taxonomy, Economic Importance, Genomics. Genetics and Genomics of Rosaceae; Folta K. M., Gardiner S. E., Eds.; Springer New York: New York, NY, 2009; pp 1–17.
-
- Potter D.; Eriksson T.; Evans R. C.; Oh S.; Smedmark J. E. E.; Morgan D. R.; Kerr M.; Robertson K. R.; Arsenault M.; Dickinson T. A.; Campbell C. S. Phylogeny and Classification of Rosaceae. Plant Syst. Evol. 2007, 266, 5–43. 10.1007/s00606-007-0539-9. - DOI
LinkOut - more resources
Full Text Sources