Multidelay ASL of the pediatric brain
- PMID: 35451851
- PMCID: PMC10996417
- DOI: 10.1259/bjr.20220034
Multidelay ASL of the pediatric brain
Abstract
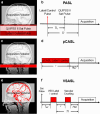
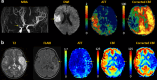
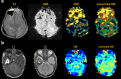
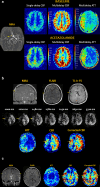
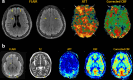
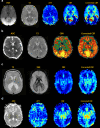
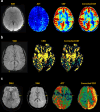
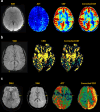
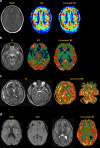
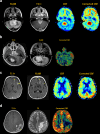
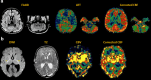
Arterial spin labeling (ASL) is a powerful noncontrast MRI technique for evaluation of cerebral blood flow (CBF). A key parameter in single-delay ASL is the choice of postlabel delay (PLD), which refers to the timing between the labeling of arterial free water and measurement of flow into the brain. Multidelay ASL (MDASL) utilizes several PLDs to improve the accuracy of CBF calculations using arterial transit time (ATT) correction. This approach is particularly helpful in situations where ATT is unknown, including young subjects and slow-flow conditions. In this article, we discuss the technical considerations for MDASL, including labeling techniques, quantitative metrics, and technical artefacts. We then provide a practical summary of key clinical applications with real-life imaging examples in the pediatric brain, including stroke, vasculopathy, hypoxic-ischemic injury, epilepsy, migraine, tumor, infection, and metabolic disease.
Figures


References
-
- Alsop DC, Detre JA, Golay X, Günther M, Hendrikse J, Hernandez-Garcia L, et al. . Recommended implementation of arterial spin-labeled perfusion MRI for clinical applications: A consensus of the ISMRM perfusion study group and the european consortium for ASL in dementia . Magn Reson Med 2015. ; 73: 102 – 16 . doi: 10.1002/mrm.25197 - DOI - PMC - PubMed

