Safety and Efficacy of the Common Vaccines against COVID-19
- PMID: 35455262
- PMCID: PMC9027683
- DOI: 10.3390/vaccines10040513
Safety and Efficacy of the Common Vaccines against COVID-19
Abstract
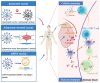
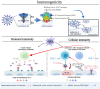
The worldwide pandemic of coronavirus disease 2019 (COVID-19) has imposed a challenge on human health worldwide, and vaccination represents a vital strategy to control the pandemic. To date, multiple COVID-19 vaccines have been granted emergency use authorization, including inactivated vaccines, adenovirus-vectored vaccines, and nucleic acid vaccines. These vaccines have different technical principles, which will necessarily lead to differences in safety and efficacy. Therefore, we aim to implement a systematic review by synthesizing clinical experimental data combined with mass vaccination data and conducting a synthesis to evaluate the safety and efficacy of COVID-19 vaccines. Compared with other vaccines, adverse reactions after vaccination with inactivated vaccines are relatively low. The efficacy of inactivated vaccines is approximately 60%, adenovirus-vectored vaccines are 65%, and mRNA vaccines are 90%, which are always efficient against asymptomatic severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection, symptomatic COVID-19, COVID-19 hospitalization, severe or critical hospitalization, and death. RNA-based vaccines have a number of advantages and are one of the most promising vaccines identified to date and are particularly important during a pandemic. However, further improvements are required. In time, all the antibody levels weaken gradually, so a booster dose is needed to maintain immunity. Compared with homologous prime-boost immunization, heterologous prime-boost immunization prompts more robust humoral and cellular immune responses.
Keywords: COVID-19; SARS-CoV-2; efficacy; prime-boost strategies; safety.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Figures

References
-
- Gomes M.G.M., Corder R.M., King J.G., Langwig K.E., Souto-Maior C., Carneiro J., Gonçalves G., Penha-Gonçalves C., Ferreira M.U., Aguas R. Individual variation in susceptibility or exposure to SARS-CoV-2 lowers the herd immunity threshold. medRxiv. 2020;540:111063. doi: 10.1016/j.jtbi.2022.111063. - DOI - PMC - PubMed
Publication types
Grants and funding
- U20A20351/National Natural Science Foundation of China
- 2021C03079/Key Research and Development Plan of Zhejiang Province
- ZDFY2020-RH-0006/Key Laboratory of Women's Reproductive Health Research of Zhejiang Province
- WKJ-ZJ-2128/key project of provincial ministry coconstruction, Health Science and Technology Project Plan of Zhejiang Province
LinkOut - more resources
Full Text Sources
Miscellaneous

