Transcriptomic Analysis of Fumarate Compounds Identifies Unique Effects of Isosorbide Di-(Methyl Fumarate) on NRF2, NF-kappaB and IRF1 Pathway Genes
- PMID: 35455458
- PMCID: PMC9026097
- DOI: 10.3390/ph15040461
Transcriptomic Analysis of Fumarate Compounds Identifies Unique Effects of Isosorbide Di-(Methyl Fumarate) on NRF2, NF-kappaB and IRF1 Pathway Genes
Abstract
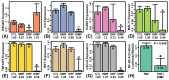
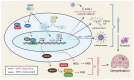
Dimethyl fumarate (DMF) has emerged as a first-line therapy for relapsing-remitting multiple sclerosis (RRMS). This treatment, however, has been limited by adverse effects, which has prompted development of novel derivatives with improved tolerability. We compared the effects of fumarates on gene expression in astrocytes. Our analysis included diroximel fumarate (DRF) and its metabolite monomethyl fumarate (MMF), along with a novel compound isosorbide di-(methyl fumarate) (IDMF). Treatment with IDMF resulted in the largest number of differentially expressed genes. The effects of DRF and MMF were consistent with NRF2 activation and NF-κB inhibition, respectively. IDMF responses, however, were concordant with both NRF2 activation and NF-κB inhibition, and we confirmed IDMF-mediated NF-κB inhibition using a reporter assay. IDMF also down-regulated IRF1 expression and IDMF-decreased gene promoters were enriched with IRF1 recognition sequences. Genes altered by each fumarate overlapped significantly with those near loci from MS genetic association studies, but IDMF had the strongest overall effect on MS-associated genes. These results show that next-generation fumarates, such as DRF and IDMF, have effects differing from those of the MMF metabolite. Our findings support a model in which IDMF attenuates oxidative stress via NRF2 activation, with suppression of NF-κB and IRF1 contributing to mitigation of inflammation and pyroptosis.
Keywords: Interferon regulatory factor; NF-kappaB; NRF2; astrocyte; dimethyl fumarate; diroximel fumarate; glial cells; multiple sclerosis; neurodegeneration; neuroinflammation.
Conflict of interest statement
WRS has received consulting reimbursement from Sytheon, Ltd., which provided funding for this study. WRS is a Symbionyx Pharmaceuticals shareholder and member of the Symbionyx advisory board. KB is CEO of Sunny BioDiscovery, Inc. (Santa Paula, CA, USA), has received consulting reimbursement from Sytheon, Ltd., and is Chief Scientific Officer of Symbionyx Pharmaceuticals. RKC is CEO and founder of Sytheon, Ltd., and Symbionyx Pharmaceuticals, with ownership interest in both companies. KB and RKC are listed as inventors of IDMF on composition of matter and application patents (US 10,597,402 and EP 3503866).
Figures







References
-
- Walton C., King R., Rechtman L., Kaye W., Leray E., Marrie R.A., Robertson N., La Rocca N., Uitdehaag B., van der Mei I., et al. Rising prevalence of multiple sclerosis worldwide: Insights from the Atlas of MS, third edition. Mult. Scler. J. 2020;26:1816–1821. doi: 10.1177/1352458520970841. - DOI - PMC - PubMed
-
- Wallin M.T., Culpepper W.J., Nichols E., Bhutta Z.A., Gebrehiwot T.T., Hay S.I., Khalil I.A., Krohn K.J., Liang X., Naghavi M., et al. Global, regional, and national burden of multiple sclerosis 1990–2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet. Neurol. 2019;18:269–285. doi: 10.1016/S1474-4422(18)30443-5. - DOI - PMC - PubMed
-
- Ortiz G.G., Pacheco-Moisés F.P., Macías-Islas M., Flores-Alvarado L.J., Mireles-Ramírez M.A., González-Renovato E.D., Hernández-Navarro V.E., Sánchez-López A.L., Alatorre-Jiménez M.A. Role of the blood-brain barrier in multiple sclerosis. Arch. Med. Res. 2014;45:687–697. doi: 10.1016/j.arcmed.2014.11.013. - DOI - PubMed
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

