A Comprehensive Review of Patented Technologies to Fabricate Orodispersible Films: Proof of Patent Analysis (2000-2020)
- PMID: 35456654
- PMCID: PMC9031760
- DOI: 10.3390/pharmaceutics14040820
A Comprehensive Review of Patented Technologies to Fabricate Orodispersible Films: Proof of Patent Analysis (2000-2020)
Abstract
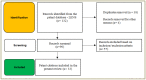
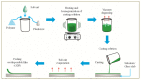
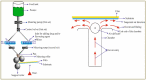
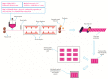
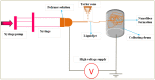
Orodispersible films (ODFs)are ultra-thin, stamp-sized, rapidly disintegrating, and attractive oral drug delivery dosage forms best suited for the pediatric and geriatric patient populations. They can be fabricated by different techniques, but the most popular, simple, and industrially applicable technique is the solvent casting method (SCM). In addition, they can also be fabricated by extrusion, printing, electrospinning, and by a combination of these technologies (e.g., SCM + printing). The present review is aimed to provide a comprehensive overview of patented technologies of the last two decades to fabricate ODFs. Through this review, we present evidence to adamantly confirm that SCM is the most popular method while electrospinning is the most recent and upcoming method to fabricate ODFs. We also speculate around the more patent-protected technologies especially in the domain of printing (two or three-dimensional), extrusion (ram or hot-melt extrusion), and electrospinning, or a combination of the methods thereof.
Keywords: electrospinning; orodispersible films; printing technologies; solvent casting.
Conflict of interest statement
The authors declare no conflict of interest.
Figures















References
-
- Behera B.K. Biopharmaceuticals: Challenges and Opportunities. CRC Press; Boca Raton, FL, USA: 2020.
-
- Ferreira A.O., Brandão M.A.F., Raposo F.J., Polonini H.C., Raposo N.R.B. Orodispersible Films for Compounding Pharmacies. Int. J. Pharm. Compd. 2017;21:454–461. - PubMed
-
- pH sensitive Compounds in Taste Masking within Oral Thin Film Strips. WO2011/072208. International Patent Publication Number No. 2011 June 16;
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources

