Effects of a Home-Based Physical Activity Programme on Blood Biomarkers and Health-Related Quality of Life Indices in Saudi Arabian Type-2 Diabetes Mellitus Patients: Protocol for a Randomised Controlled Trial
- PMID: 35457335
- PMCID: PMC9030925
- DOI: 10.3390/ijerph19084468
Effects of a Home-Based Physical Activity Programme on Blood Biomarkers and Health-Related Quality of Life Indices in Saudi Arabian Type-2 Diabetes Mellitus Patients: Protocol for a Randomised Controlled Trial
Abstract
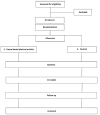
The Kingdom of Saudi Arabia is renowned for its high incidence of type-2 diabetes mellitus, with a prevalence rate of around 33%, which is expected to increase to 45.8% by 2030. Engagement in regular physical activity has been shown to significantly attenuate non-communicable diseases including type-2 diabetes. However, the overall rate of physical inactivity among Saudi Arabian adults is currently 80.5%, owing to time pressures, high-density traffic, poor air quality, lack of suitable exercise places/sports facilities, lack of social/friends support, gender, cultural barriers, low self-confidence, lack of time and environmental factors. Previous analyses have shown that home-based activity interventions can be effective. Therefore, given the aforementioned barriers to physical activity in Saudi Arabia; a home-based physical activity may be an ideal solution in type-2 diabetic patients. This manuscript describes the study protocol for a randomized control trial, examining the effects of a home-based physical activity intervention in Saudi Arabian adults with type-2 diabetes. The study will recruit 62 individuals with type-2 diabetes from the Jazan region of the Kingdom of Saudi Arabia, who will be individually randomized to either a physical activity or control group. This 24-week investigation will involve 12-weeks of physical activity in the physical activity group and feature three examination points i.e., baseline, 12-weeks and 24-weeks (follow-up). The primary study outcome is the between-group difference in blood HbA1c levels relative to controls. Secondary outcomes measures will be between-group differences in anthropometric, blood lipid, physical fitness, and patient-reported quality of life outcomes pertinent to type-2 diabetes. Statistical analysis will be conducted on an intention-to-treat basis. The trial has been granted ethical approval by Jazan University, Health Research Ethics Committee (REF: 2177) and formally registered as a trial (NCT04937296). We expect dissemination of the study findings from this investigation to be through publication in a leading peer-reviewed journal.
Keywords: HbA1c; Saudi Arabia; diabetes mellitus; exercise; physical activity.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
References
-
- Robertson R.P., Bogardus C. Antagonist: Diabetes and insulin resistance: Philosophy, science, and the multiplier hypothesis. Discussion. J. Lab. Clin. Med. 1995;125:560–565. - PubMed
-
- Blas E., Kurup A.S. Equity, Social Determinants and Public Health Programmes. World Health Organization; Geneva, Switzerland: 2010.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical