Reticulon-1A mediates diabetic kidney disease progression through endoplasmic reticulum-mitochondrial contacts in tubular epithelial cells
- PMID: 35469894
- PMCID: PMC9329239
- DOI: 10.1016/j.kint.2022.02.038
Reticulon-1A mediates diabetic kidney disease progression through endoplasmic reticulum-mitochondrial contacts in tubular epithelial cells
Abstract
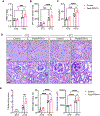
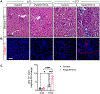
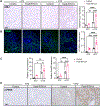
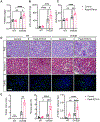
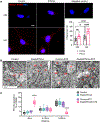
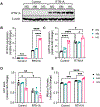
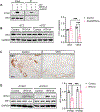
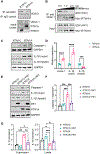
Recent epidemiological studies suggest that some patients with diabetes progress to kidney failure without significant albuminuria and glomerular injury, suggesting a critical role of kidney tubular epithelial cell (TEC) injury in diabetic kidney disease (DKD) progression. However, the major risk factors contributing to TEC injury and progression in DKD remain unclear. We previously showed that expression of endoplasmic reticulum-resident protein Reticulon-1A (RTN1A) increased in human DKD, and the increased RTN1A expression promoted TEC injury through endoplasmic reticulum (ER) stress response. Here, we show that TEC-specific RTN1A overexpression worsened DKD in mice, evidenced by enhanced tubular injury, tubulointerstitial fibrosis, and kidney function decline. But RTN1A overexpression did not exacerbate diabetes-induced glomerular injury or albuminuria. Notably, RTN1A overexpression worsened both ER stress and mitochondrial dysfunction in TECs under diabetic conditions by regulation of ER-mitochondria contacts. Mechanistically, ER-bound RTN1A interacted with mitochondrial hexokinase-1 and the voltage-dependent anion channel-1 (VDAC1), interfering with their association. This disengagement of VDAC1 from hexokinase-1 resulted in activation of apoptotic and inflammasome pathways, leading to TEC injury and loss. Thus, our observations highlight the importance of ER-mitochondrial crosstalk in TEC injury and the salient role of RTN1A-mediated ER-mitochondrial contact regulation in DKD progression.
Keywords: diabetic kidney disease; endoplasmic reticulum stress; endoplasmic reticulum-mitochondrial contacts; kidney tubular epithelial cells.
Published by Elsevier Inc.
Conflict of interest statement
Figures

References
-
- Collins AJ, Foley RN, Herzog C, et al. US Renal Data System 2010 Annual Data Report. Am J Kidney Dis 57: A8, e1–526. - PubMed
-
- Mauer M, Zinman B, Gardiner R, et al. ACE-I and ARBs in early diabetic nephropathy. Journal of the renin-angiotensin-aldosterone system : JRAAS 2002; 3: 262–269. - PubMed
-
- Garofolo M, Russo E, Miccoli R, et al. Albuminuric and non-albuminuric chronic kidney disease in type 1 diabetes: Association with major vascular outcomes risk and all-cause mortality. J Diabetes Complications 2018; 32: 550–557. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials

