Blockade of inhibitory killer cell immunoglobulin-like receptors and IL-2 triggering reverses the functional hypoactivity of tumor-derived NK-cells in glioblastomas
- PMID: 35474089
- PMCID: PMC9042843
- DOI: 10.1038/s41598-022-10680-4
Blockade of inhibitory killer cell immunoglobulin-like receptors and IL-2 triggering reverses the functional hypoactivity of tumor-derived NK-cells in glioblastomas
Abstract
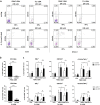
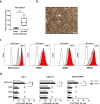
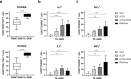
Killer cell immunoglobulin-like receptors (KIRs) comprise a group of highly polymorphic inhibitory receptors which are specific for classical HLA class-I molecules. Peripheral blood and freshly prepared tumor cell suspensions (n = 60) as well as control samples (n = 32) were investigated for the distribution, phenotype, and functional relevance of CD158ab/KIR2DL1,-2/3 expressing NK-cells in glioblastoma (GBM) patients. We found that GBM were scarcely infiltrated by NK-cells that preferentially expressed CD158ab/KIR2DL1,-2/3 as inhibitory receptors, displayed reduced levels of the activating receptors CD335/NKp46, CD226/DNAM-1, CD159c/NKG2C, and showed diminished capacity to produce IFN-γ and perforin. Functional hypoactivity of GBM-derived NK-cells persisted despite IL-2 preactivation. Blockade with a specific KIR2DL-1,2/3 monoclonal antibody reversed NK-cell inhibition and significantly enhanced degranulation and IFN-γ production of IL-2 preactivated NK-cells in the presence of primary GBM cells and HLA-C expressing but not HLA class-I deficient K562 cells. Additional analysis revealed that significant amounts of IL-2 could be produced by tumor-derived CD4+ and CD8+CD45RA- memory T-cells after combined anti-CD3/anti-CD28 stimulation. Our data indicate that both blockade of inhibitory KIR and IL-2 triggering of tumor-derived NK-cells are necessary to enhance NK-cell responsiveness in GBM.
© 2022. The Author(s).
Conflict of interest statement
C.S., J.W., M.H, B.B., W.S. and C.T. declare that they have no conflict of interest. A.S.M. received research support and travel expenses from Novartis. H.W. received compensation for serving on scientific advisory boards/steering committees and as a consultant from Bayer Healthcare, Biogen, Sanofi Genzyme, Merck Serono, Roche, and Novartis. He has received speaker honoraria and travel support from Bayer Vital GmbH, Bayer Schering AG, Biogen, CSL Behring, EMD Serono, Fresenius Medical Care, Genzyme, Merck Serono, Omniamed, Novartis, and Sanofi Aventis. H.W. also received research support from Bayer Healthcare, Bayer Vital, Biogen, Merck Serono, Novartis, Sanofi Genzyme, Sanofi US and Teva Pharma, Merck Serono, and Novartis. O.M.G. received speaker honoraria and travel expenses from Roche, Bristol- Myers Squibb and MagForce. He received compensation as a consultant from Gilead Sciences and Bayer AG.
Figures


References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

