Clerodane Diterpenes from Casearia corymbosa as Allosteric GABAA Receptor Modulators
- PMID: 35475609
- PMCID: PMC9150179
- DOI: 10.1021/acs.jnatprod.1c00840
Clerodane Diterpenes from Casearia corymbosa as Allosteric GABAA Receptor Modulators
Abstract
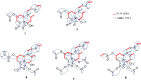
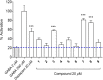
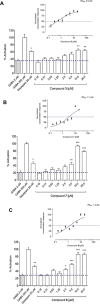
An EtOAc extract of Casearia corymbosa leaves led to an allosteric potentiation of the GABA signal in a fluorometric imaging plate reader (FLIPR) assay on Chinese hamster ovary (CHO) cells stably expressing GABAA receptors with an α1β2γ2 subunit composition. The activity was tracked by HPLC-based activity profiling, and four known (2, 3, 4, and 8) and five new clerodane-type diterpenoids (1, 5-7, and 9) were isolated. Compounds 1-8 were obtained from the active time window. The absolute configuration of all compounds was established by ECD. Compounds 3, 7, and 8 exhibited EC50 values of 0.5, 4.6, and 1.4 μM, respectively. To explore possible binding sites at the receptor, the most abundant diterpenoid 8 was tested in combination with diazepam, etazolate, and allopregnanolone. An additive potentiation of the GABA signal was observed with these compounds, while the effect of 8 was not inhibited by flumazenil, a negative allosteric modulator at the benzodiazepine binding site. Finally, the activity was validated in voltage clamp studies on Xenopus laevis oocytes transiently expressing GABAA receptors of the α1β2γ2S and α1β2 subtypes. Compound 8 potentiated GABA-induced currents with both receptor subunit compositions [EC50 (α1β2γ2S) = 43.6 μM; Emax = 809% and EC50 (α1β2) = 57.6 μM; Emax = 534%]. The positive modulation of GABA-induced currents was not inhibited by flumazenil, thereby confirming an allosteric modulation independent of the benzodiazepine binding site.
Conflict of interest statement
The authors declare no competing financial interest.
Figures



References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Chemical Information

