Rigid 3D-spiro chromanone as a crux for efficient antimicrobial agents: synthesis, biological and computational evaluation
- PMID: 35478839
- PMCID: PMC9034028
- DOI: 10.1039/d1ra03497a
Rigid 3D-spiro chromanone as a crux for efficient antimicrobial agents: synthesis, biological and computational evaluation
Abstract
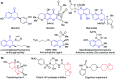
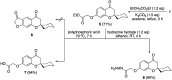
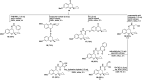
The development of new and effective antimicrobial agents with novel chemical skeletons and working mechanisms is highly desirable due to the increased number of resistant microbes. Different new compounds based upon a 3D-spiro chromanone scaffold such as Mannich bases 2 and 3 in addition to azo dye 4 were synthesized. Besides, the condensation reactions of the hydrazide-spiro chromanone 8 with different ketonic reagents led to the synthesis of pyrazoles (9 & 10) and anils (11 & 13). Moreover, the methoxyl substituted spiro chromanone 14 was condensed with different hydrazines and hydrazides to give the corresponding hydrazones 15-18 in up to 85% yields. The condensation of the hydrazone 18 with salicylaldehyde yielded coumarinyl spiro chromanone 19 in an excellent yield, whereas its reaction with benzaldehyde followed by hydrazine afforded aminopyrazole derivative 21 in 82% yield. The antimicrobial evaluation suggested that hydrazide 8 has a substantial activity against different microbes (S. aureus: D = 22 mm, MIC = 1.64 μM; E. coli: D = 19 mm, MIC = 1.64 μM; C. albicans: D = 20 mm, MIC = 6.57 μM). Moreover, promising antimicrobial activities were observed for azo dye 4 (D = 13-19 mm, MIC = 5.95-11.89 μM), hydrazone 17 (D = 17-23 mm, MIC = 1.88-3.75 μM), and aminopyrazole 21 (D = 14-19 mm, MIC = 2.24-8.98 μM). The molecular docking revealed that compounds 4, 8, 17, and 21 had good to high binding affinities with different microbial targets such as penicillin-binding proteins (-7.4 to -9.9 kcal), DNA gyrase (-7.8 to -9.0 kcal), lanosterol 14-alpha demethylase (-8.2 to -11.2 kcal), and exo-beta-1,3-glucanase (-8.2 to -11.9 kcal). The QSAR analysis ascertained a good correlation between the antimicrobial activity of 3D-spiro chromanone derivatives and their structural and/or physicochemical parameters.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
The authors declare no competing financial interests.
Figures




Similar articles
-
New Hydrazides and Hydrazide-Hydrazones of 2,3-Dihalogen Substituted Propionic Acids: Synthesis and in vitro Antimicrobial Activity Evaluation.Chem Biodivers. 2017 Aug;14(8). doi: 10.1002/cbdv.201700075. Epub 2017 Jun 26. Chem Biodivers. 2017. PMID: 28444991
-
Synthesis, computational docking and molecular dynamics studies of a new class of spiroquinoxalinopyrrolidine embedded chromanone hybrids as potent anti-cholinesterase agents.RSC Adv. 2024 Jun 12;14(26):18815-18831. doi: 10.1039/d4ra02432j. eCollection 2024 Jun 6. RSC Adv. 2024. PMID: 38867740 Free PMC article.
-
Structure based design and synthesis of 3-(7-nitro-3-oxo-3,4-dihydroquinoxalin-2-yl)propanehydrazide derivatives as novel bacterial DNA-gyrase inhibitors: In-vitro, In-vivo, In-silico and SAR studies.Bioorg Chem. 2022 Dec;129:106186. doi: 10.1016/j.bioorg.2022.106186. Epub 2022 Oct 5. Bioorg Chem. 2022. PMID: 36215786
-
Quinoline Hydrazide/Hydrazone Derivatives: Recent Insights on Antibacterial Activity and Mechanism of Action.ChemMedChem. 2023 Mar 1;18(5):e202200571. doi: 10.1002/cmdc.202200571. Epub 2023 Jan 20. ChemMedChem. 2023. PMID: 36617503 Review.
-
Chromanone-A Prerogative Therapeutic Scaffold: An Overview.Arab J Sci Eng. 2022;47(1):75-111. doi: 10.1007/s13369-021-05858-3. Epub 2021 Jun 30. Arab J Sci Eng. 2022. PMID: 34226859 Free PMC article. Review.
Cited by
-
Antimicrobial Activity and DFT Studies of a Novel Set of Spiropyrrolidines Tethered with Thiochroman-4-one/Chroman-4-one Scaffolds.Molecules. 2022 Jan 18;27(3):582. doi: 10.3390/molecules27030582. Molecules. 2022. PMID: 35163847 Free PMC article.
References
-
- Ellis G. P., Chromenes, chromanones and chromones, John Willy & Sons, New York, 1977
LinkOut - more resources
Full Text Sources

