Evolutionary origin and sequence signatures of the heterodimeric ABCG5/ABCG8 transporter
- PMID: 35481657
- PMCID: PMC8994503
- DOI: 10.1002/pro.4297
Evolutionary origin and sequence signatures of the heterodimeric ABCG5/ABCG8 transporter
Abstract
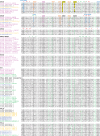
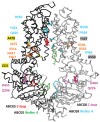
ATP-binding cassette (ABC) systems, characterized by ABC-type nucleotide-binding domains (NBDs), play crucial roles in various aspects of human physiology. Human ABCG5 and ABCG8 form a heterodimeric transporter that functions in the efflux of sterols. We used sequence similarity search, multiple sequence alignment, phylogenetic analysis, and structure comparison to study the evolutionary origin and sequence signatures of ABCG5 and ABCG8. Orthologs of ABCG5 and ABCG8, supported by phylogenetic analysis and signature residues, were identified in bilaterian animals, Filasterea, Fungi, and Amoebozoa. Such a phylogenetic distribution suggests that ABCG5 and ABCG8 could have originated in the last common ancestor of Amorphea (the unikonts), the eukaryotic group including Amoebozoa and Opisthokonta. ABCG5 and ABCG8 were missing in genomes of various lineages such as snakes, jawless vertebrates, non-vertebrate chordates, echinoderms, and basal metazoan groups. Amino-acid changes in key positions in ABCG8 Walker A motif and/or ABCG5 C-loop were observed in most tetrapod organisms, likely resulted in the loss of ATPase activity at one nucleotide-binding site. ABCG5 and ABCG8 in Ecdysozoa (such as insects) exhibit elevated evolutionary rates and accumulate various changes in their NBD functional motifs. Alignment inspection revealed several residue positions that show different amino-acid usages in ABCG5/ABCG8 compared to other ABCG subfamily proteins. These residues were mapped to the structural cores of transmembrane domains (TMDs), the NBD-TMD interface, and the interface between TMDs. They serve as sequence signatures to differentiate ABCG5/ABCG8 from other ABCG subfamily proteins, and some of them may contribute to substrate specificity of the ABCG5/ABCG8 transporter.
Keywords: ABC systems; ABC transporters; ABCG5; ABCG8; sequence signatures; sterol efflux.
© 2022 The Protein Society.
Conflict of interest statement
None declared.
Figures


Similar articles
-
Association of ABCG5 and ABCG8 Transporters with Sitosterolemia.Adv Exp Med Biol. 2024;1440:31-42. doi: 10.1007/978-3-031-43883-7_2. Adv Exp Med Biol. 2024. PMID: 38036873 Review.
-
Functional asymmetry of nucleotide-binding domains in ABCG5 and ABCG8.J Biol Chem. 2006 Feb 17;281(7):4507-16. doi: 10.1074/jbc.M512277200. Epub 2005 Dec 12. J Biol Chem. 2006. PMID: 16352607
-
Transcriptional response of zebrafish larvae exposed to lindane reveals two detoxification genes of ABC transporter family (abcg5 and abcg8).Comp Biochem Physiol C Toxicol Pharmacol. 2020 Jun;232:108755. doi: 10.1016/j.cbpc.2020.108755. Epub 2020 Mar 31. Comp Biochem Physiol C Toxicol Pharmacol. 2020. PMID: 32244031
-
Transmembrane Polar Relay Drives the Allosteric Regulation for ABCG5/G8 Sterol Transporter.Int J Mol Sci. 2020 Nov 19;21(22):8747. doi: 10.3390/ijms21228747. Int J Mol Sci. 2020. PMID: 33228147 Free PMC article.
-
Snapshots of ABCG1 and ABCG5/G8: A Sterol's Journey to Cross the Cellular Membranes.Int J Mol Sci. 2022 Dec 28;24(1):484. doi: 10.3390/ijms24010484. Int J Mol Sci. 2022. PMID: 36613930 Free PMC article. Review.
References
-
- Dassa E, Bouige P. The ABC of ABCS: A phylogenetic and functional classification of ABC systems in living organisms. Res Microbiol. 2001;152(3–4):211–229. - PubMed
-
- Dermauw W, Van Leeuwen T. The ABC gene family in arthropods: Comparative genomics and role in insecticide transport and resistance. Insect Biochem Mol Biol. 2014;45:89–110. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

