Optoelectronic Neural Interfaces Based on Quantum Dots
- PMID: 35482955
- PMCID: PMC9100496
- DOI: 10.1021/acsami.1c25009
Optoelectronic Neural Interfaces Based on Quantum Dots
Abstract
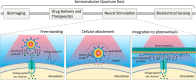
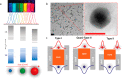
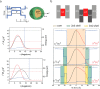
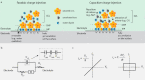
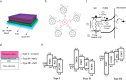
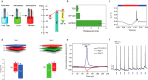
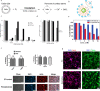
Optoelectronic modulation of neural activity is an emerging field for the investigation of neural circuits and the development of neural therapeutics. Among a wide variety of nanomaterials, colloidal quantum dots provide unique optoelectronic features for neural interfaces such as sensitive tuning of electron and hole energy levels via the quantum confinement effect, controlling the carrier localization via band alignment, and engineering the surface by shell growth and ligand engineering. Even though colloidal quantum dots have been frontier nanomaterials for solar energy harvesting and lighting, their application to optoelectronic neural interfaces has remained below their significant potential. However, this potential has recently gained attention with the rise of bioelectronic medicine. In this review, we unravel the fundamentals of quantum-dot-based optoelectronic biointerfaces and discuss their neuromodulation mechanisms starting from the quantum dot level up to electrode-electrolyte interactions and stimulation of neurons with their physiological pathways. We conclude the review by proposing new strategies and possible perspectives toward nanodevices for the optoelectronic stimulation of neural tissue by utilizing the exceptional nanoscale properties of colloidal quantum dots.
Keywords: nanocrystal; neural interface; neural stimulation; optoelectronics; quantum dot.
Conflict of interest statement
The authors declare no competing financial interest.
Figures




References
-
- Lozano A. M.; Lipsman N.; Bergman H.; Brown P.; Chabardes S.; Chang J. W.; Matthews K.; McIntyre C. C.; Schlaepfer T. E.; Schulder M.; Temel Y.; Volkmann J.; Krauss J. K. Deep Brain Stimulation: Current Challenges and Future Directions. Nat. Rev. Neurol. 2019, 15 (3), 148–160. 10.1038/s41582-018-0128-2. - DOI - PMC - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources

