Engineering encapsulated ionic liquids for next-generation applications
- PMID: 35492767
- PMCID: PMC9043619
- DOI: 10.1039/d1ra05034f
Engineering encapsulated ionic liquids for next-generation applications
Abstract
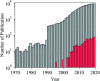
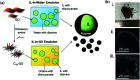
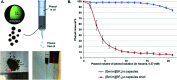
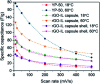
Ionic liquids (ILs) have attracted considerable attention in several sectors (from energy storage to catalysis, from drug delivery to separation media) owing to their attractive properties, such as high thermal stability, wide electrochemical window, and high ionic conductivity. However, their high viscosity and surface tension compared to conventional organic solvents can lead to unfavorable transport properties. To circumvent undesired kinetics effects limiting mass transfer, the discretization of ILs into small droplets has been proposed as a method to increase the effective surface area and the rates of mass transfer. In the present review paper, we summarize the different methods developed so far for encapsulating ILs in organic or inorganic shells and highlight characteristic features of each approach, while outlining potential applications. The remarkable tunability of ILs, which derives from the high number of anions and cations currently available as well as their permutations, combines with the possibility of tailoring the composition, size, dispersity, and properties (e.g., mechanical, transport) of the shell to provide a toolbox for rationally designing encapsulated ILs for next-generation applications, including carbon capture, energy storage devices, waste handling, and microreactors. We conclude this review with an outlook on potential applications that could benefit from the possibility of encapsulating ILs in organic and inorganic shells.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
The authors declare no competing financial interests.
Figures





References
-
- MacFarlane D. R., Kar M. and Pringle J. M., An Introduction to Ionic Liquids, in Fundamentals of Ionic Liquids, Wiley-VCH Verlag GmbH & Co. KGaA, 2017, pp. 1–25, 10.1002/9783527340033.ch1 - DOI
-
- Earle M. J. Seddon K. R. Ionic Liquids. Green Solvents for the Future. Pure Appl. Chem. 2000;72(7):1391–1398. doi: 10.1351/pac200072071391. - DOI
-
- Matic A. Scrosati B. Ionic Liquids for Energy Applications. MRS Bull. 2013;38(7):533–537. doi: 10.1557/mrs.2013.154. doi: 10.1557/mrs.2013.154. - DOI
Publication types
LinkOut - more resources
Full Text Sources

