MOF-Zn-NHC as an efficient N-heterocyclic carbene catalyst for aerobic oxidation of aldehydes to their corresponding carboxylic acids via a cooperative geminal anomeric based oxidation
- PMID: 35492781
- PMCID: PMC9043340
- DOI: 10.1039/d1ra05494e
MOF-Zn-NHC as an efficient N-heterocyclic carbene catalyst for aerobic oxidation of aldehydes to their corresponding carboxylic acids via a cooperative geminal anomeric based oxidation
Abstract
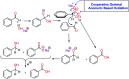
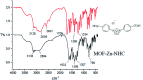
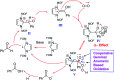
As an efficient heterogenous N-heterocyclic carbene (NHC) catalyst, MOF-Zn-NHC was used in the aerobic oxidation of aryl aldehydes to their corresponding carbocyclic acids via an anomeric based oxidation. Features such as mild reaction conditions and no need for a co-catalyst or oxidative reagent can be considered as the major advantages of the presented method in this study.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
The authors declare no competing financial and non-financial interests.
Figures

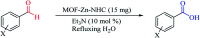

Similar articles
-
N-Heterocyclic Carbene/Carboxylic Acid Co-Catalysis Enables Oxidative Esterification of Demanding Aldehydes/Enals, at Low Catalyst Loading.Angew Chem Int Ed Engl. 2021 Sep 1;60(36):19631-19636. doi: 10.1002/anie.202104712. Epub 2021 Jul 20. Angew Chem Int Ed Engl. 2021. PMID: 34010504 Free PMC article.
-
Sequential Connection of Mutually Exclusive Catalytic Reactions by a Method Controlling the Presence of an MOF Catalyst: One-Pot Oxidation of Alcohols to Carboxylic Acids.Inorg Chem. 2020 Dec 7;59(23):17573-17582. doi: 10.1021/acs.inorgchem.0c02809. Epub 2020 Nov 20. Inorg Chem. 2020. PMID: 33216548
-
Synthesis of Thioesters from Aldehydes via N-Heterocyclic Carbene (NHC) Catalyzed Radical Relay.Chemistry. 2023 Mar 22;29(17):e202203716. doi: 10.1002/chem.202203716. Epub 2023 Feb 16. Chemistry. 2023. PMID: 36583288
-
N-Heterocyclic Carbene (NHC)/Metal Cooperative Catalysis.Top Curr Chem (Cham). 2019 Nov 13;377(6):35. doi: 10.1007/s41061-019-0266-z. Top Curr Chem (Cham). 2019. PMID: 31720874 Review.
-
Carboxylation Reactions with Carbon Dioxide Using N-Heterocyclic Carbene-Copper Catalysts.Chem Rec. 2020 Jun;20(6):494-512. doi: 10.1002/tcr.201900060. Epub 2019 Oct 1. Chem Rec. 2020. PMID: 31573147 Review.
Cited by
-
Application of Zr-MOFs based copper complex in synthesis of pyrazolo[3, 4-b]pyridine-5-carbonitriles via anomeric-based oxidation.Sci Rep. 2023 Jun 9;13(1):9388. doi: 10.1038/s41598-023-34172-1. Sci Rep. 2023. PMID: 37296128 Free PMC article.
-
Catalytic Application of Functionalized Bimetallic-Organic Frameworks with Phosphorous Acid Tags in the Synthesis of Pyrazolo[4,3-e]pyridines.ACS Omega. 2023 Jul 3;8(28):25303-25315. doi: 10.1021/acsomega.3c02580. eCollection 2023 Jul 18. ACS Omega. 2023. PMID: 37483221 Free PMC article.
-
Metallosalen modified carbon nitride a versatile and reusable catalyst for environmentally friendly aldehyde oxidation.Sci Rep. 2024 Apr 11;14(1):8498. doi: 10.1038/s41598-024-58946-3. Sci Rep. 2024. PMID: 38605107 Free PMC article.
-
Catalytic application of a bimetallic-organic framework with phosphorus acid groups in the preparation of pyrido[2,3-d]pyrimidines via cooperative vinylogous anomeric-based oxidation.RSC Adv. 2025 Apr 2;15(13):10150-10163. doi: 10.1039/d4ra08430f. eCollection 2025 Mar 28. RSC Adv. 2025. PMID: 40176821 Free PMC article.
-
Facile synthesis of polyoxometalate supported on magnetic graphene oxide as a hybrid catalyst for efficient oxidation of aldehydes.Sci Rep. 2022 Nov 2;12(1):18491. doi: 10.1038/s41598-022-21991-x. Sci Rep. 2022. PMID: 36323774 Free PMC article.
References
-
- Song H. Kim Y. Park J. Kim K. Lee E. Synlett. 2016;27:477–485.
-
- Land M. A. Clyburne J. A. Synlett. 2016;27:2416–2424. doi: 10.1055/s-0035-1562622. - DOI
LinkOut - more resources
Full Text Sources
Research Materials

