Effect of the Nipple-Excising Breast-Conserving Therapy in Female Breast Cancer: A Competing Risk Analysis and Propensity Score Matching Analysis of Results Based on the SEER Database
- PMID: 35494069
- PMCID: PMC9048049
- DOI: 10.3389/fonc.2022.848187
Effect of the Nipple-Excising Breast-Conserving Therapy in Female Breast Cancer: A Competing Risk Analysis and Propensity Score Matching Analysis of Results Based on the SEER Database
Abstract
Introduction: Due to the lack of randomized controlled trial, the effectiveness and oncological safety of nipple-excising breast-conserving therapy (NE-BCT) for female breast cancer (FBC) remains unclear. We aimed to explore and investigate the prognostic value of NE-BCT versus nipple-sparing breast-conserving therapy (NS-BCT) for patients with early FBC.
Methods: In this cohort study, data between NE-BCT and NS-BCT groups of 276,661 patients diagnosed with tumor-node-metastasis (TNM) stage 0-III FBC from 1998 to 2015 were retrieved from the Surveillance, Epidemiology, and End Results database. Propensity score matching analysis, Kaplan-Meier, X-tile, Cox proportional hazards model, and competing risk model were performed to evaluate the effectiveness and oncological safety for patients in NE-BCT and NS-BCT groups.
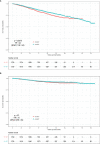
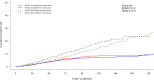
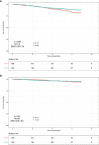
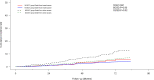
Results: A total of 1,731 (0.63%) patients received NE-BCT (NE-BCT group) and 274,930 (99.37%) patients received NS-BCT (NS-BCT group); 44,070 subjects died after a median follow-up time of 77 months (ranging from 1 to 227 months). In the propensity score matching (PSM) cohort, NE-BCT was found to be an adversely independent prognostic factor affecting overall survival (OS) [hazard ratio (HR), 1.24; 95% CI, 1.06-1.45, p=0.0078]. Subjects in NE-BCT group had similar breast-cancer-specific survival (BCSS) (HR, 1.15; 95%CI, 0.88-1.52, p=0.30) and worse other-causes-specific death (OCSD) (HR, 1.217; 95%CI, 1.002-1.478, p=0.048<0.05) in comparison with those in the NS-BCT group.
Conclusions: Our study demonstrated that the administration of NE-BCT is oncologically safe and reliable and can be widely recommended in clinics for women with non-metastatic breast cancer.
Keywords: SEER database; breast-conserving therapy; competing risk model; female breast cancer; nipple areola complex.
Copyright © 2022 Li, Zhao, Yan, Yang, Qiu, Chen, Zhou, Niu, Yan, Zhang, Zhang, He and Zhou.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


References
-
- Gradishar WJ, Moran MS, Abraham J, Aft R, Agnese D, Allison KH, et al. . Version 4. 2021, NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines). In: Breast Cancer Version2. Plymouth: National Comprehensive Cancer Network (2021). Available at: www.nccn.org/patients.
-
- Fisher B, Anderson S, Bryant J, Margolese RG, Deutsch M, Fisher ER, et al. . Twenty-Year Follow-Up of a Randomized Trial Comparing Total Mastectomy, Lumpectomy, and Lumpectomy Plus Irradiation for the Treatment of Invasive Breast Cancer. N Engl J Med (2002) 347(16):1233–41. doi: 10.1056/NEJMoa022152 - DOI - PubMed
-
- Voogd AC, van Oost FJ, Rutgers EJ, Elkhuizen PH, van Geel AN, Scheijmans LJ, et al. . Dutch Study Group on Local Recurrence After Breast Conservation (BORST Group). Long-Term Prognosis of Patients With Local Recurrence After Conservative Surgery and Radiotherapy for Early Breast Cancer. Eur J Cancer (2005) 41(17):2637–44 doi: 10.1016/j.ejca.2005.04.040 - DOI - PubMed
-
- Didier F, Radice D, Gandini S, Bedolis R, Rotmensz N, Maldifassi A, et al. . Does Nipple Preservation in Mastectomy Improve Satisfaction With Cosmetic Results, Psychological Adjustment, Body Image and Sexuality? Breast Cancer Res Treat (2009) 118(3):623–33. doi: 10.1007/s10549-008-0238-4 - DOI - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

